Lymphedema after treatment of breast cancer: a comprehensive review
Abstract
Lymphedema is a chronic and morbid complication that can result from breast cancer treatment involving surgery and/or radiation therapy. Breast cancer related lymphedema (BCRL) can result in functional and psychological problems that can affect a patient’s quality of life. Lymphedema is a pathologic condition of the lymphatic system in which protein-containing fluid accumulates in the interstitial tissue leading to tissue inflammation, fibrosis, and adipose hypertrophy. Clinical manifestations of lymphedema include swelling, induration, skin changes, and decreased functionality of the affected limb. A common risk factor associated with BCRL is the management of the axilla. Advances in research have shown sentinel lymph biopsy to be equally effective as axillary lymph node dissection in staging the axilla, in appropriate cases. In addition, with breast-conserving therapy it has been shown that radiation therapy also increases the risk of BCRL. Overall, approximately 1 in 6 women treated for breast cancer will develop lymphedema within months to years after diagnosis and treatment. Lymphedema is a progressive disease with a subclinical phase that can last for years before becoming clinically evident. While management of breast cancer has improved, it is important that we develop a coordinated and standardized approach to better diagnose, assess, and manage BCRL. This includes patient education, a thorough physical examination, access to confirmatory testing when necessary, a conservative multimodality treatment plan that includes compression and decongestive therapy, and being able to offer operative management when conservative management is ineffective.
Keywords
Introduction
It has been estimated that in 2018, there were over 2 million new diagnoses of breast cancer worldwide[1]. The American Cancer Society reported an estimated 252,710 new cases of invasive breast cancer diagnoses among women and 2,470 cases in men in 2017. In the United States, the lifetime risk of being diagnosed with breast cancer increased from 1 in 11, in the 1970s, to 1 in 12, currently, partly due to increased detection in screening, hormone replacement therapy after menopause, the rising prevalence of obesity, and changes in reproductive patterns[2]. By 2026, estimated breast cancer survivors were predicted to reach over 4 million, and 45% of them would be older than age 50 years[3]. Treatment of breast cancer includes surgery, chemotherapy, endocrine therapy, and radiation therapy but there are some adverse events related to these treatments. One of these complications is lymphedema of the upper extremity, a chronic condition, which can be a challenge to both patients and clinicians. Various studies demonstrate a wide variety of incidence rates in which approximately 16.6% of breast cancer survivors develop lymphedema[4]. This variation depends on the extent of breast and axillary surgery as well as the use of adjuvant radiation therapy[5,6].
Lymphedema is defined as a set of pathophysiologic conditions in which protein-containing fluid accumulates in soft tissues due to an interruption of lymphatic flow which can result in inflammation, fibrosis, and hypertrophy of adipose tissue. It is a chronic disease that can be difficult to treat and is often associated with both physical and psychologic morbidity. Due to the increased incidence of breast cancer diagnoses, some requiring axillary lymph node surgery and/or radiation, it is important that we understand the possible prevention, management, and early detection of BCRL to reduce the degree of morbidity associated with BCRL. There is a continued effort in research and new advances in the field to improve management of BCRL.
Methods
We performed PubMed/MEDLINE and Cochrane searches of the English-language literature. Studies of all dates were included to provide adequate background as well as to capture new studies that have emerged in the past 5 years. General keywords used for this search were lymphedema, breast-cancer related lymphedema, decongestive lymphatic therapy, lymphedema management, and axillary lymph node surgery. Published guidelines and resources from the International Society of Lymphology, the National Comprehensive Cancer Network, the American Society of Clinical Oncology, the American Society of Breast Surgeons, and the American Physical Therapy Association, were also included for review. In addition, relevant publications were manually reviewed for additional resources relevant to this review.
Pathophysiology
The underlying mechanism of lymphedema is dysfunction in the lymphatic transport system. Three primary functions of the lymphatic system include maintaining fluid balance, serving as a nutritional adjunct, and aiding in host defense against disease[7,8]. The lymphatic system maintains fluid balance by removing excess fluid from capillaries that accumulates in the interstitium in order to maintain the steady state of the interstitial pressure. Lymphatic fluid, known as lymph, is drained by blind-ended lymphatic capillaries. It is then filtered through lymph nodes and ultimately reenters the circulatory system, via the thoracic duct, where the peripheral venous blood enters the right atrium of the heart[9]. Under normal conditions, the same amount is transported to the interstitium as is transported from it, a balance that is disrupted in lymphedema due to reduced lymph transport capacity, thus leading to fluid accumulation and swelling. Disruption of this system subsequently leads to a decrease in oxygen tension resulting in reactive tissue fibrosis and chronic tissue inflammation. In addition, the buildup of protein in the interstitium leads to an increased osmotic pressure which drives fluid into the interstitium causing edema and the clinical manifestations of lymphedema[9,10].
According to traditional definitions, lymphedema is divided into two types: primary and secondary. Primary lymphedema is often classified according to the age at which the edema first appeared: congenital lymphedema, (present at birth), lymphedema praecox (onset at time of puberty or as late as third decade of life), and lymphedema tarda (typically appears after age 35)[11]. Secondary lymphedema is due to an extrinsic disruption of, or obstruction of the lymphatic system due to trauma. The forms of trauma include surgery to the axilla, radiation therapy, chemotherapy, or inflammation and scarring from metastasis to the lymph nodes[8,10]. More recently, Damstra et al.[12] reports that as all interstitial fluid is transported by lymphatics, the overload of fluid can be relative, based on lymphatic impairment and dysfunction of the lymphatic system (afterload impairment), or absolute, based on increased filtration rate (preload is raised). Under this classification, the traditional categories of primary and secondary lymphedema would be considered afterload impairment.
In the United States, secondary lymphedema is most commonly due to malignancy or its related therapy[10]. These malignancies include, but are not limited to, breast cancer, melanoma, and lymphoma. Breast cancer treated with surgery and/or radiation accounts for the majority of upper-extremity lymphedema. Studies have demonstrated BCRL rates of 24% to 49% after mastectomy and 4% to 28% after lumpectomy, with a history of axillary lymph node surgery[13-18].
Lymphatic drainage of the breast
One of the most common causes of upper extremity lymphedema is breast cancer and its related treatment. The axilla has approximately 20 to 30 lymph nodes, but this can vary. The axilla receives drainage from 75% of the ipsilateral breast, regions of the upper back and shoulder, lower neck, the chest and the upper anterolateral abdominal wall[19]. These nodes are further divided into 6 groups: axillary vein (lateral), external mammary, scapular and subscapular, central, subclavicular, and interpectoral groups. Surgeons typically define the axillary lymph nodes in relation to the pectoralis minor muscle: levels I, II, and III-lateral to, posterior to, and medial to the pectoralis minor muscle, respectively.
Breast cancer related lymphedema
Breast cancer, depending on the stage and other factors, can be managed surgically with or without involvement of the axillary lymph nodes. Axillary lymph node surgery includes ALND or SLNB. ALND typically involves removal of level I and II nodes whereas SLNB involves removing those lymph nodes identified as the first node(s) to receive lymphatic drainage from the site of the tumor and should be the first site of lymphatic spread[20].
Incidence and risk factors
It has been posited that SLNB confers less morbidity than ALND and was determined to provide accurate staging of the axillary nodal basin. Larson and colleagues found a correlation between the risk of lymphedema formation and the number of removed lymph nodes; when greater than 10 nodes were removed, the risk of edema was 28% vs. 9% if 1 to 10 nodes were removed[21]. In addition, the literature demonstrates an average risk of lymphedema with ALND to be 28%, with a range of 11% to 57%[22]. This variation in incidence over many large trials is secondary to the method of detection of lymphedema, how closely lymphedema is monitored, surgical technique, and length of follow-up[21-23]. Smaller studies have shown that the number of positive lymph nodes, postoperative radiation therapy, extent of surgery, obesity, and other patient characteristics contribute to the risk of lymphedema and thus the varying rates of incidence[23-25]. The Axillary Lymphatic Mapping Against Nodal Axillary Clearance trial demonstrated that lymphedema was observed in approximately 5% of patients who had SLNB vs. 13% of those who had ALND[26]. The National Surgical Breast and Bowel Project (NSABP-32) randomized 5,400 patients to SLNB followed by ALND vs. SLNB alone demonstrating an 8% risk of BCRL with SLNB[19,27]. The American College of Surgeons Oncology Group organized a prospective international, multicenter study, trial Z0010, in which upper extremity edema was demonstrated in 7% of patients with SLNB, performed by a wide range of surgeons (including, but not limited to, level of experience)[28]. Overall, many randomized controlled trials (from 2002 to 2014), have demonstrated decreased incidence of BCRL after SLNB vs. ALND, with follow-up ranging from 12 months to 60 months[29-37].
In 2007, Thompson et al.[38] introduced the technique of axillary reverse mapping (ARM) to describe the drainage of the arm within the axilla to facilitate reducing the risk of lymphedema after axillary lymph node surgery. The technique involves preoperative injection of technetium sulfur colloid into the subareolar plexus along with intradermal injection of blue dye into the ipsilateral upper extremity. Over the years, studies have demonstrated that identifying the lymphatic drainage using ARM helped to reduce the risk of disruption of lymphatics and subsequent BCRL, when combined with axillary lymph node surgery[38-40].
In addition to axillary lymph node surgery being a risk factor for BCRL, it has been shown that radiation therapy is an independent risk factor for the development of BCRL with reported rates of 2% to 5%, even in the absence of lymph node surgery[41,42]. Target areas for radiation therapy depends on many factors, including but not limited to, type of surgery, lymph node involvement and patient characteristics such as age and co-morbidities. These target areas include the whole breast, partial breast, and regional lymph nodes. Whole breast radiation therapy (WBRT) generally includes treatment of the entire breast with modifications of treatment field boundaries based on whether a patient has had mastectomy versus lumpectomy. Partial breast radiation is focused therapy to a limited area of the breast and is delivered as accelerated partial breast irradiation. Regional node irradiation (RNI), is therapy to specific regional node basins alone or in conjunction with WBRT depending on the extent of disease. The European Organization for Research and Treatment of Cancer after Mapping of the Axilla: Radiotherapy or Surgery randomized controlled trial demonstrated a 13% risk of lymphedema in those with ALND and WBRT vs. 5% for SLNB, WBRT, and RNI[43]. It also showed that axillary radiotherapy provides comparable axillary control compared to completion ALND after a positive SLNB. The National Cancer Institute of Canada Clinical Trials Group conducted a study which demonstrated a BCRL incidence of 4.5% after ALND and WBRT vs. 8.4% after ALND, WBRT and RNI[44]. Radiation to the axilla is associated with 2 to 4.5 times greater risk of lymphedema and 8 to 10 times greater risk when a patient receives ALND and radiation treatment[45-47].
In addition to the known risk factors for BCRL, there are conflicting studies about adjuvant and neoadjuvant chemotherapy as possible risk factors for BCRL. Some studies indicate that adjuvant chemotherapy is a potential risk factor for BCRL[4,48-53]. DiSipio et al.[4] did a meta-analysis of 72 studies published from 2000 to 2012 to assess the incidence rates of BCRL, and a meta-analysis of 29 studies published from 2000 to 2012, to assess the risk factors. This study demonstrated that about 75% of the studies reviewed indicated chemotherapy as a risk factor for developing BCRL[4]. Kilbreath et al.[48] did a recent large, prospective cohort study, using objective limb measurements which showed that patients who had taxane-based chemotherapy were at risk for arm swelling at 6 months and 12 months; those that experience arm swelling at 6 and 12 months were at greater risk for BCRL at 18 months (6 months OR: 5.6; 95% CI: 2.0-16.9) (12 months OR: 13.5; 95% CI: 4.8-38.1). These findings were also supported by Zhu et al.[52] who also found a correlation between taxane-based adjuvant chemotherapy and the cumulative incidence of BCRL. Conversely, Swaroop et al.[54] did not find any correlation between taxane-based chemotherapy and the development of BCRL, however there was some correlation between docetaxol to be a risk factor for mild swelling compared to no chemotherapy and non-taxane based chemotherapy. While there are new studies looking at neoadjuvant therapy as a risk factor for the development of BCRL, the findings are inconclusive. Specht et al.[55] showed that there was an increased risk of BCRL in patients found to have residual nodal disease after neoadjuvant chemotherapy. However, at this time more research needs to be done further assess the effects of neoadjuvant chemotherapy as a risk factor.
A well-studied, independent risk factor for BCRL is high BMI, defined as BMI > 30 kg/m2[35,50,56-60]. Jamallo et al.[62] and other smaller studies have also correlated this finding that women with BMI of 30 kg/m2 or greater, were at least 3.6 times more likely to develop BCRL[53,61]. Furthermore, Jamallo et al.[62] found that post-operative weight fluctuations greater than 10 pounds per month, either lost or gained, increased BCRL[53]. With this information, counseling patients about healthy weight, as well managing weight fluctuations, can contribute to helping to decrease the incidence of BCRL with this well-known, modifiable risk factor.
Clinical presentation
The clinical manifestations of lymphedema are secondary to an inflammatory response to the chronic accumulation of protein-containing interstitial fluid and adipose tissue. Lymph stasis, or decreased flow, has been shown to contribute to lipogenesis and fat deposition which later leads to increased fibrocyte activation and connective tissue growth[63-65].
Many rating scales exist to characterize the clinical progression of lymphedema. The International Society of Lymphology has developed a staging system to classify lymphedema based on the physical appearance of the limb [Table 1][66]. Within each stage, severity based on limb volume difference can be further characterized as minimal (< 20% increase), moderate (20%-40%), or severe (> 40%) increase[20]. A more recent classification system developed by Campisi et al.[67] uses clinical presentation as well as lymphoscintigraphy findings to classify lymphedema[Table 1][8]. Additionally, Chang et al.[68] incorporates ICG lymphangiography to classify lymphedema which further aids in surgical planning in lymphedema of the upper extremity [Table 1].
| Stage | International society of lymphology | Campisi et al.[67] | Chang et al.[68] |
|---|---|---|---|
| 0 | Latent or subclinical Patients complain of heavy sensation and/or numbness in arm May exist for months to years before overt edema occurs | ||
| 1 | Early accumulation of protein-rich fluid May have soft, pitting edema: limb elevation leads to complete resolution of swelling No fibrosis | A. No clinical edema despite the presence of lymphatic dysfunction as demonstrated on lymphoscintigraphy
B. Mild edema that spontaneously regresses with elevation Limb excess volume: 0%-20% | Many patent lymphatic vessels, with minimal, patchy dermal backflow |
| 2 | Limb elevation alone rarely reduces swelling Fibrosis present: reduces ability of skin to indent with pressure | Persistent edema that regresses only partially with elevation Limb excess volume: 21%-40% | Moderate number of patent lymphatic vessels, with segmented dermal backflow |
| 3 | Lymphostatic elephantiasis: no pitting edema present Severe fibrosis and hypertrophic skin changes such as hyperkeratosis, fat deposits and warty outgrowths | Persistent, progressive edema; recurrent erysipeloid lymphangitis Limb excess volume: 41%-60% | Few patient lymphatic vessels, with extensive dermal backflow involving the entire arm |
| 4 | Fibrotic lymphedema with column limb Limb excess volume: > 60% | No patent lymphatic vessels seen, with severe dermal backflow involving the entire arm and extending to the dorsum of the hand | |
| 5 | Elephantiasis with severe limb deformation, including scleroinductive pachydermitis and widespread lymphostatic warts Limb excess volume: > 60% |
Diagnosis
Diagnosis of BCRL is based on consideration of risk factors, associated symptoms, and clinical signs and is typically made during physical examination. Clinically evident lymphedema will present with various levels of pitting edema, which can involve any aspect of the upper extremity, usually first noticeable in the digits of the hand. Affected patients may complain of increased heaviness and decreased activity of the affected limb. Because BCRL can develop months to years after onset of lymphatic insult, it is important that patients are assessed for this during follow-up appointments even when there is no complaint of BCRL symptoms. Literature has demonstrated that most women present with BCRL within the first two years of diagnosis and treatment[69-71]. A study of 1,713 women who underwent breast-conserving therapy showed that 40% of them who presented with arm edema had mild edema at diagnosis[72]. While BCRL is the likely diagnosis, when evaluating women treated for breast cancer, there are other causes of limb swelling, such as other malignancy, upper extremity deep venous thrombosis, and infection that must also be considered. Therefore, if indicated, it is important to carry out appropriate studies, to exclude these diagnoses.
The literature has described a wide variety of noninvasive methods for evaluating limb volume when lymphedema is clinically evident. Options include bioelectrical impedance analysis (BIA), tape measurement, perometry, and water displacement[73]. BIA determines the opposition to flow of an electric current (electrical impedance) through body tissue which is then used to estimate total body water[74]. The resistance of flow of electric current is due to differences in conductivity of different tissues. Based on these factors, the device is able to selectively measure water content without quantifying adipose or fibrous tissues[75-77]. Studies have demonstrated that the results of BIA are affected by body temperature and body hydration status so using it may not always be accurate[78].
A common method used to diagnose upper-extremity edema is circumferential and volumetric measurement using anatomical landmarks[79]. Affected and unaffected arm circumferences are measured sequentially at 4 points: the metacarpal-phalangeal joints, the wrist, 10 cm distal to the lateral epicondyles, and 15 cm proximal to the lateral epicondyles. Differences of 2 cm or more, or if converted to volume, 10% or a 200 mL increase, at any point compared with the contralateral arm are considered by some experts to be clinically significant[80,81]. This method requires consideration of patient factors such as baseline discrepancies between dominant and nondominant limbs, muscle mass differences, body habitus, and post-treatment changes that may occur. Because it has been found that pre-treatment sequential measurements may prove to be clinically meaningful in order to have a baseline to improve accuracy of post-treatment assessment and diagnosis, newer methods have emerged to more accurately classify BCRL. It has been shown that quantification methods that include absolute volume changes to diagnosis BCRL are inaccurate and vary from patient to patient. The arm size of patients diagnosed with breast cancer have been shown to vary within a broad range[82]. Volume change correlates with preoperative arm volume, patient weight, and BMI however relative volume change (RVC) is independent of patient weight. In order to move towards creating a standardized classification of BCRL, RVC would be a more appropriate measure than absolute volume change[83]. Furthermore, for patients undergoing unilateral breast surgery, Ancukiewicz et al.[84] established a method for measuring RVC by considering the pre-operative and post-operative follow-up ipsilateral arm volumes and the pre-operative and post-operative follow-up volumes on the contralateral arms. This method uses the contralateral arm as a control and assumes any weight gained by the patient is distributed equally between the arms[82,84]. This method established by Ancukiewicz et al.[84], however, cannot be applied to women undergoing bilateral mastectomy. A revised version of this model, established by Miller et al.[85] includes the patient’s weight at each arm measurement (pre-operative and post-operative measurements). As assumed by the unilateral method, Miller et al.[85] demonstrated a 1:1 linear relationship between change in patient weight and volume of the contralateral arm in those undergoing unilateral breast surgery, which lead to the proposal of a weight-adjusted formula to quantify arm volume in patients undergoing bilateral breast surgery.
Furthermore, the National Cancer Institute, which has standardized reporting of adverse events in clinical trials classifies levels of lymphedema based on percentage interlimb volume/circumference discrepancies, under the Common Terminology Criteria for Adverse Events, v4.0 (CTCAE)[69]:
Level 1: 5%-10%;
Level 2: > 10%-30%;
Level 3: > 30% - Lymphorrhea and gross anatomic deviation from normal contour may occur. This level interferes with activities of daily living;
Level 4: Although rare, the progression to malignancy, such as lymphangiosarcoma, can occur, for which limb amputation may be warranted. In other classification systems, this level may also be referred to as “end-stage” lymphedema.
The CTCAE criteria offers both subjective (interlimb discrepancy) and objective measures to accurately classify lymphedema which offers a more standardized assessment of lymphedema.
It is known that there is a subclinical phase of BCRL and recent studies of this phase have emerged. Specht et al.[86]did a prospective study that screened 1,173 patients with perometry and found that low-level volume changes (RVC of > 3% and < 5%, and > 5% and < 10%) within 3 months of surgery were significantly associated with development of BCRL[86]. For volume changes of RVC of > 5% and < 10% were significantly associated with development of BCRL greater than 3 months post-operatively, while an RVC of > 3% and < 5% within 3 months of surgery was not associated with BCRL development[86]. Findings from this study suggest that patients with an RVC from > 3% to < 5% within 3 months of surgery and > 5% to < 10% at any point should be monitored closely with clinical exams and are candidates for early intervention if warranted. For suspected cases of subclinical BCRL, based on abovementioned risk factors, additional tests can be done to confirm the diagnosis.
Radionuclide imaging of the lymphatic system, lymphoscintigraphy, may be necessary although it is not a widely used technique[86]. Lymphoscintigraphy is a relatively noninvasive technique which involves an intradermal injection of radiolabeled colloid in the distal aspect of the affected limb followed by imaging of the lymphatic vasculature[10,70]. This method can delineate the lymphatic anatomy and function. Common abnormal findings include absent or delayed radiotracer transport and poorly visualized lymphatic collectors and lymph nodes[71].
Computed tomography (CT) and magnetic resonance imaging (MRI) can also be used to delineate lymphatic dysfunction and can complement findings of lymphoscintigraphy. These imaging modalities can also be used alone if lymphoscintigraphy is not available. CT imaging has been shown to have a 97% sensitivity (SN) and a 100% specificity (SP) in confirming the diagnosis of lymphedema. MRI has been shown to have superior soft tissue imaging than CT, it offers better detail of lymphatic anatomy, and has equivalent SN and SP[10,87]. MRI findings associated with lymphedema include, “honeycombing” of the subcutaneous tissue due fibrotic tissue and fluid accumulation surrounding adipose tissue, epifascial fluid lakes, and the absence of edema within muscle compartments[88-91].
In addition to current modalities for detecting and diagnosing BCRL, recent studies have explored the utility and efficacy of using indocyanine green (ICG) to improve the performance of sentinel lymph node mapping. Struk et al.[92] performed a prospective trial of a cohort of 198 consecutive early breast cancer patients eligible for sentinel lymph node biopsy to assess the value of the combination of ICG and methylene blue (MB) dye in patients undergoing SLNB. The nodal detection rate of ICG, MB, and ICG with MB samples was 97, 89, and 99.5% (P < 0.001), respectively, with the combination method yielding a superior identification result[92]. The addition of ICG to the MB method resulted in the identification of more lymph nodes (median 3 vs. 2) and more positive axillae (22.7% involved axillae were discovered by fluorescence only) than either method alone[92]. This study concluded that ICG with MB dual tracing modality, without involvement of radioactive isotopes, exhibits great potential as an alternative to traditional standard mapping methods[92]. This study gives insight into ICG with MB as a viable alternative for accurately mapping the lymphatics prior to axillary lymph node surgery. Furthermore, the findings of this study, along with other studies have shown that the use of ICG lymphography is an additional, reliable method to evaluate the upper extremity lymphatic drainage of those patients with clinically evident BCRL[93-97]. ICG lymphography can detect lymphatic channels up to 2 cm deep from the surface of the skin and can show lymphatic channels, lymphatic valves, and lymphatic flow immediately after injection of ICG[98]. The ICG lymphography patterns are of two types: linear and dermal backflow (DB), with linear patterns defining normal lymphatic flow and DB patterns defining extremity lymphedema. DB is further classified according to severity of the lymphedema[97,98].
The key to timely and accurate diagnosis of BCRL requires thorough history-taking and physical examination, early detection and coordination of the care team, which can be supplemented with various studies and imaging modalities in cases of subclinical BCRL.
Treatment
Conservative management
The initial treatment of clinically evident BCRL should be conservative (non-operative). The mainstay of conservative therapy relies on the finding that reduction of pitting edema can be achieved with compression. This often involves multilayer inelastic lymphedema bandaging or controlled compression therapy, where the compression garment’s size is reduced regularly as the swelling decreases[99,100]. It has been shown that both of these methods alone significantly reduce excess edema volume by as much as 31% and 46%, respectively[100,101].
Many devices have been created to provide massage through mechanical or compressional means that do not require experienced personnel[102]. The principle of these machines is to provide a pumping-like action with the goal of improving lymph and venous circulation to the affected limb. A review of the effectiveness of pneumatic compression therapy devices did not show a significant improvement when compared to the standard management of lymphedema[102].
Complete decongestive therapy (CDT), first described in the 1800s, is considered the standard management of lymphedema. It is more labor-intensive than the abovementioned therapies and relies on principles of skin hygiene, limb compression, and exercise[19,103]. Vodder further demonstrated that the methods of CDT would augment lymphatic contractility, increase lymphatic flow, and reduce lymphatic fluid from accumulating in the affected limb[104]. CDT involves 2 phases of physical therapy. Phase 1 is typically 4 weeks and consists of skin care, manual lymphatic drainage, compression bandaging and specialized physical therapy exercise with subsequent lymphatic massage. Phase 2 involves applying low resistance short-stretch compressive bandages in a multi-layered fashion with the aim of enhancing lymphatic drainage[19,103,104]. A review of the literature, including randomized controlled trials, has shown that CDT leads to an average 50% reduction in excess volume in patients with pitting edema[105-107]. A systemic review of 26 studies of CDT also demonstrated decreased limb volume and improved quality of life[108]. However, some small randomized-controlled trials have shown no significant difference in extremity volume reduction when comparing CDT to compression garments over 4 to 6 weeks of therapy[100,109,110]. When patients were followed for a year, the lack of significant difference still existed[110]. Despite the finding of these smaller studies, CDT remains the standard therapy for lymphedema and has been found to be effective. A drawback of CDT is that it requires multiple health care providers and is labor-intensive, where maintaining the overall benefits depends largely on patient compliance and ability to continue required exercises and therapies.
Aquatic lymphatic therapy (ALT) is another treatment modality for BCRL under the direction of a physical therapist[111-113]. ALT is based on the principles of the intrinsic properties of water including buoyant force, water viscosity, and hydrostatic pressure combined with compression therapy. It is done weekly, in a group setting and the patient is actively involved in their treatment. A recent systemic review of ALT did not show a significant benefit of ALT over standard land-based care for improving lymphedema status or physical function in people with upper extremity lymphedema[113].
Pharmacologic agents have been studied for treatment of chronic lymphedema. This includes benzopyrones because they cause phagocytosis and proteolysis by macrophages which results in improved lymph flow and regulation of tissue osmolarity[114-116]. The resulting degradation of the excess protein decreases the oncotic pressure of the interstitium causing reduction in tissue edema. A Cochrane review of 15 randomized controlled trials could not conclusively comment on the effectiveness of benzopyrenes in the management of lymphedema[117].
In addition to the various conservative therapy options, adequate patient education regarding activity levels, exercise, infection prophylaxis, and treatment expectations is important. BCRL is a chronic disease that has been shown to require a multidisciplinary and multimodal approach to management in order to be maximally successful. Patients must also understand that these therapies will alleviate their symptoms but are not curative. Thus, active patient participation and compliance with treatment plans is important.
Operative management
In selected patients with long-term complications of BCRL or those for whom an adequate trial of non-operative management has failed, operative management can be considered. Surgical options include resection, microsurgery, tissue-transfer, and liposuction.
The underlying principles of resection involves removing subcutaneous tissue to the level of the underlying fascia, with or without removal of the skin. While this does not improve, or directly address the underlying lymphatic dysfunction, it serves to improve patient comfort level and increase functionality of the affected limb[118]. As a result, the lymphedema may return. In addition, the cosmetic outcome is usually not favorable. Thus, it is important to inform patients of the risks and benefits of excisional procedures.
Alternatively, microsurgery is an option which involves directly correcting the underlying lymphatic dysfunction. In the literature, interposition autologous lymphatic-venous-lymphatic anastomoses has been described[119]. This procedure involves inserting large autologous venous grafts between lymphatic collectors above and below the site of obstruction to lymphatic flow[19,119]. Alternative anastomotic procedures exist and it has been shown that these procedures result in a mean volume reduction of 69% and an 87% reduction in the incidence of cellulitis[119]. However, the outcomes could not be adequately assessed due to missing information regarding pre-operative factors such as limb volume and the use of compression garments. It has been shown in other studies that when microsurgery is used in combination with compression therapy, specifically elastic bandaging, there was a decrease in arm volume between 22% and 30%[120,121]. The drawback of adequately analyzing the direct benefit of stand-alone microsurgical technique is that patients used in these studies were all instructed to wear continuous compression garments post-operatively.
Because of the need for continuous compression garments, the popularity of tissue transfer for refractory lymphedema has been increasing. This technique involves harvesting groin nodes and reimplanting them in the axilla or hand. Many small retrospective and prospective studies have shown a volume reduction of 22% to 81%[121-128].
Liposuction, also referred to as suction-assisted lipectomy, is based on the principle of fat hypertrophy that occurs as sequelae of long-standing lymphedema. This technique involves circumferential liposuction from hand to shoulder. The arm is then bandaged with compression to control post-operative bleeding and edema[129]. In addition, the patient wears a compression garment and the arm is elevated for up to 4 days. The patient is then closely followed with controlled compression therapy which is key to the post-operative management and the limb is assessed yearly. One study has shown no recurrences after a 15-year follow-up[129]. Other prospective studies have demonstrated a reduction in lymphedema ranging from 103% to 123%[130-132].
Conclusion
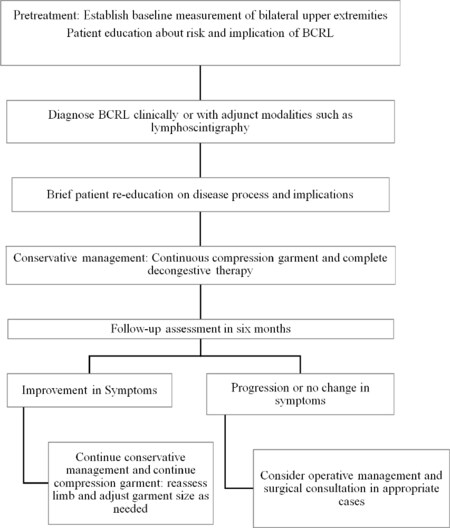
Many factors are involved with the increased incidence of breast cancer that is seen in the United States. As a result, breast cancer patients will be in the care of many providers while undergoing treatment. It is important that they are all aware of BCRL and the most effective way of screening and management of this chronic and sometimes debilitating disease. A thorough history and physical examination is the mainstay of appropriately addressing this disease. Clinical suspicion must be followed through with appropriate testing to confirm a diagnosis of BCRL and then appropriate follow-up and reassessment [Figure 1].
In addition, patients must be well-educated by clinicians on the risk of lymphedema, and its implications prior to undergoing treatment. This allows patients to be well-informed, better equipped to participate in their care and assist with early detection and management of BCRL.
Over the years, landmark trials have changed and improved the way breast cancer is managed with better overall outcomes. This includes the way the axilla is approached with regards to surgery, chemotherapy, and radiation therapy. Although the literature demonstrates a decreased risk of BCRL after SLNB, when compared to ALND, the incidence of BCRL remains significant and is a chronic, and sometimes debilitating disease, that warrants continued research into improving detection, management, and overall outcomes. A standardized, multidisciplinary approach is warranted in order to effectively treat and control this chronic condition that is sometimes not detected until years after a patient has been diagnosed and treated for breast cancer.
Declarations
Authors’ contributionsMade substantial contributions to conception and design of the study and performed data analysis and interpretation: Ayre K, Parker C
Performed data acquisition, as well as provided administrative, technical, and material support: Ayre K, Parker C
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Authors 2019.
REFERENCES
1. Bray F, Ferlay J, Soerjomataram I, Siegel R, Torre L, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence of and mortality worldwide for 36 cancers in 185 countries. Ca Cancer J Clin 2018;68:394-424.
2. American Cancer Society. Breast cancer facts and figures 2017-2018. Available from: https://www.cancer.org/research/cancer-facts-statistics/breast-cancer-facts-figures.html [Last accessed on 23 Dec 2018].
3. Tandra P, Kallam A, Krishnamurthy J. Identification and management of lymphedema in patients with breast cancer. J Oncol Pract 2019;5:255-62.
4. DiSipio T, Rye S, Newman B, Hayes S. Incidence of unilateral arm lymphoedema after breast cancer: a systematic review and meta-analysis. Lancet Oncol 2013;14:500-15.
5. Erickson VS, Pearson MI, Ganz PA, Adams J, Kahn KL. Arm edema in breast cancer patients. J Natl Cancer Inst 2001;93:96-111.
6. Lee TS, Kilbreath SL, Refshauge KM, Herbert RD, Beith JM. Prognosis of the upper limb following surgery and radiation for breast cancer:11. Lymphedema. CMAJ. 2001;164:191-9. Available from: http://www.cmaj.ca/content/164/2/191 [Last accessed on 15 Dec 2018].
7. Mortimer PS, Rockson SG. New developments in clinical aspects of lymphatic disease. J Clin Invest 2014;124:915-21.
8. Garza R 3rd, Skoracki R, Hock K, Povoski S. A comprehensive overview on the surgical management of secondary lymphedema of the upper and lower extremities related to prior oncologic therapies. BMC Cancer 2017;17:468-85.
9. Grada AA, Philips TJ. Lymphedema: pathophysiology and clinical manifestations. J Am Acad Dermatol 2017;77:1009-20.
10. Warren AG, Brorson H, Borud LJ, Slavin AS. Lympedema: a comprehensive review. Ann Plast Surg 2007;59:464-72.
12. Damstra RJ, Halk A. The Dutch lymphedema guidelines based on the international classification of functioning, disability, and health and the chronic care model. J Vasc Surg Venous Lymphat Disord 2017;5:756-65.
13. Petrek JA, Senie RT, Peters M, Rosen PP. Lymphedema in a cohort of breast carcinoma survivors 20 years after diagnosis. Cancer 2001;92:1368-77.
14. Querci della Rovere G, Ahmad I, Singh P, Ashley S, Daniels IR, et al. An audit of the incidence of arm lymphoedema after prophylactic level I/II axillary dissection without division of the pectoralis minor muscle. Ann R Coll Surg Engl 2003;85:158-61.
16. Hinrichs CS, Watroba NL, Rezaishiraz H, Giese W, Hurd T, et al. Lymphedema secondary to postmastectomy radiation: incidence and risk factors. Ann Surg Oncol 2004;11:573-80.
17. Coen JJ, Taghian AG, Kachnic LA, Assaad SI, Powell SN. Risk of lymphedema after regional nodal irradiation with breast conservation therapy. Int J Radiat Oncol Biol Phys 2003;55:1209-15.
18. Werner RS, McCormick B, Petrek J, Cox L, Cirrincione C, et al. Arm edema in conservatively managed breast cancer: obesity is a major predictive factor. Radiology 1991;180:177-84.
19. Rivere AE, Klimberg VS. Lymphedema in the postmastectomy patient: pathophysiology, prevention, and management. In: Bland KI, Copeland EM, Klimberg VS, Gradishar WJ, editors. The Breast: Comprehensive management of benign and malignant disease. Philadelphia: Elselvier; 2018. pp. 514-30.
20. Purushotham AD, Upponi S, Klevasath MB, Bobrow L, Millar K, et al. Morbidity after sentinel lymph node biopsy in primary breast cancer: results from a randomized controlled trial. J Clin Oncol 2005;23:4312-21.
21. Larson D, Weinstein M, Goldberg I, Silver B, Recht A, et al. Edema of the arm as a function of the extent of axillary surgery in patients with stage I-II carcinoma of the breast treated with primary radiotherapy. Int J Rad Oncol Biol Phys 1986;12:1575-82.
22. Shaitelman SF, Cromwell KD, Rasmussen JC, Stout NL, Armer JM, et al. Recent pro¬gress in the treatment and prevention of cancer-related lymph¬edema. CA Cancer 2015;65:55-81.
23. Hayes SC, Janda M, Cornish B, Battistutta D, Newman B. Lymph¬edema after breast cancer: incidence, risk factors and effect of upper body. J Clin Oncol 2008;26:3536-42.
24. Albert US, Koller M, Kopp I, Lorenz W, Schulz KD, et al. Early self-reported impairments in arm functioning of primary breast cancer patients predict late side effects of axillary lymph node dissection: results from a population-based cohort study. Breast Cancer Res Treat 2006;100:285-92.
25. Deutsch M, Land S, Begovic M, Sharif S. The incidence of arm edema in women with breast cancer randomized on the National Surgical Adjuvant Breast and Bowel Project study B-04 to radical mastectomy versus total mastectomy and radiotherapy versus total mastectomy alone. Int J Radiat Oncol Biol Phys 2008;70:1020-1024.
26. Mansel RE, Fallowfield L, Kissin M, Goyal A, Newcombe RG, et al. Randomized multicenter trial of sentinel node biopsy versus standard axillary treatment in operable breast cancer: The ALMANAC Trial. J Natl Cancer Inst 2006;98:599-609.
27. Ashikaga T, Krag DN, Land SR, Julian TB, Anderson SJ, et al. Morbidity results from the NSABP B-32 trial comparing sentinel lymph node dissection versus axillary dissection. J Surg Oncol 2010;102:111-118.
28. Wilke LG, McCall LM, Posther KE, Whitworth PW, Reintgen DS, et al. Surgical complications associated with sentinel lymph node biopsy: results from a pro-spective international cooperative group trial. Ann Surg Oncol 2006;13:491-500.
29. Haid A, Koberle-Wuhrer R, Knauer M, Burtscher J, Fritzsche H, et al. Morbidity of breast cancer patients following complete axillary dissection or sentinel node biopsy only: a comparative evaluation. Breast Cancer Res Treat 2002;73:31-36.
30. Blanchard DK, Donohue JH, Reynolds C, Grant CMD. Relapse and morbidity in patients undergoing sentinel lymph node biopsy alone or with axillary dissection for breast cancer. Arch Surg 2003;138:482-488.
31. Ronka R, Smitten K, Tasmuth T, Leidenius M. One-year mor¬bidity after sentinel node biopsy and breast surgery. Breast 2005;14:28-36.
32. Francis WP, Abghari P, Du W, Rymal C, Suna M, et al. Improving surgical outcomes: standardizing the reporting of incidence and severity of acute lymphedema after sentinel lymph node biopsy and axillary lymph node dissection. Am J Surg 2006;192:636-9.
33. Lucci A, McCall LM, Beitsch PD, Whitworth PW, Reintgen DS, et al; American College of Surgeons Oncology Group. Surgical complications associated with sentinel lymph node dissection (SLND) plus axillary lymph node dissection compared with SLND alone in the American College of Surgeons Oncology Group Trial Z0011. J Clin Oncol 2007;25:3657-63.
34. Langer I, Guller U, Berclaz G, Koechli OR, Schaer G, et al. Morbidity of sentinel lymph node biopsy (SLN) alone versus SLN and completion axillary lymph node dissection after breast cancer surgery: a prospective Swiss multi-center study on 659 patients. Ann Surg 2007;245:452-61.
35. McLaughlin SA, Wright MJ, Morris KT, Sampson MR, Brockway JP, et al. Prevalence of lymph¬edema in women with breast cancer 5 years after sentinel lymph node biopsy or axillary dissection: patient perceptions and precau¬tionary behaviors. Clin Oncol 2008;10:5220-56.
36. Sagen A, Kaaresen R, Sandvik L, Thune I, Risberg MA. Upper limb physical function and adverse effects after breast cancer surgery: a prospective 2.5-year follow-up study and preoperative measures. Arch Phys Med Rehabil 2014;95:875-81.
37. Sackey H, Magnuson A, Sandelin K, Liljegren G, Bergkvist L, et al. Arm lymphoedema after axillary surgery in women with invasive breast cancer. Br J Surg 2014;101:390-7.
38. Thompson M, Kororuian S, Henry-Tillman R, Adkins L, Mumford S, et al. Axillary reverse mapping (ARM): a new concept to identify and enhance lymphatic preservation. Ann Surg Oncol 2007;14:1890-15.
39. Boneti C, Korourian S, Bland K, Cox K, Adkins LL, et al. Axillary reverse mapping: mapping and preserving arm lymphatics may be important in preventing lymphedema during sentinel node biopsy. J Am Coll Surg 2009;206:1038-42.
40. Tummel E, Ochoa D, Korourian S, Betzold R, Adkins L, et al. Does Axillary Reverse Mapping Prevent Lymphedema After Lymphadenectomy? Ann Surg 2017;265:987-92.
41. Kiel KD, Rademacker AW. Early stage breast cancer: arm edema after wide excision and breast irradiation. Radiology 1996;198:279-83.
42. Johansen J, Overgaard J, Blichert-Toft M, Overgaard M. Treatment morbidity associated with the management of the axilla in breast-conserving therapy. Acta Oncol 2000;39:349-54.
43. Donker M, van Tienhoven G, Straver ME, Meijnen P, van de Velde CJ, et al. Radiotherapy or surgery of the axilla after a positive sentinel node in breast cancer (EORTC 10981-22023 AMAROS): a randomised, mul¬ticentre, open-label, phase 3 non-inferiority trial. Lancet Oncol 2014;15:1303-10.
44. Whelan TJ, Olivotto IA, Parulekar WR, Ackerman I, Chua BH, et al. Regional nodal irra¬diation in early-stage breast cancer. NCIC-CTG MA.20 107107. N Engl J Med 2015;373:307-16.
45. Kissin M, Querci Della Rovere G, Easton D, Westbury G. Risk of lymphedema following treatment of breast cancer. Br J Surg 1986;73:580-4.
46. Kwan W, Jackson J, Weir LM, Dingee C, McGregor G, et al. Chronic arm morbidity after curative breast cancer treatment: Prevalence and impact on quality of life. J Clin Oncol 2002;20:4242-8.
47. Silberman H. Axillary lymphadenectomy for breast-cancer: impact on survival. In: Silberman H, Silberman AW, editors. Surgical Oncology: Multidisciplinary Approach to Difficult Problems. London: Arnold; 2002. pp. 369-85.
48. Kilbreath SL, Refshauge KM, Beith JM, Ward LC, Ung OA, et al. Risk factors for lymphoedema in women with breast cancer: A large prospective cohort. Breast 2016;28:29-36.
49. Kim M, Kim SW, Lee SU, Lee MK, Jung SY, et al. A model to estimate the risk of breast cancer-related lymphedema: Combinations of treatment-related factors of the number of dissected axillary lymph nodes, adjuvant chemotherapy, and radiation therapy. Int J Radiat Oncol Biol Phys 2013;86:498-503.
50. Ahmed RL, Schmitz KH, Prizment AE, Folsom AR. Risk factors for lymphedema in breast cancer survivors, the Iowa Women’s Health Study. Breast Cancer Res Treat 2011;130:981-91.
51. Jung SY, Shin KH, Kim M, Chung SH, Lee S, et al. Treatment factors affecting breast cancer-relatede lymphedema after systemic chemotherapy and radiotherapy in stage II/III breast cancer patients. Breast Cancer Res Treat 2014;148:91-8.
52. Zhu W, Li D, Li X, Ren J, Chen W, et al. Association between adjuvant docetaxel-based chemotherapy and breast cancer-related lymphedema. Anticancer Drugs 2017;28:350-55.
53. Gillespie TC, Sayegh HE, Brunelle CL, Daniell KM, Taghian AG. Breast cancer-related lymphedema: risk factors, precautionary measures, and treatments. Gland Surg 2018;7:379-403.
54. Swaroop N, Ferguson CM, Horick NK, Skolny MN, Miller CL, et al. Impact of adjuvant taxane-based chemotherapy on development of breast cancer-related lymphedema: results from a large prospective cohort. Breast Cancer Res Treat 2015;151:393-403.
55. Specht MC, Miller CL, Skolny MN, Jammallo LS, O’Toole J, et al. Residual lymph node disease after neoadjuvant chemotherapy predicts an increased risk of lymphedema in node-positive breast cancer patients. Ann Surg Oncol 2013;20:2835-41.
56. Crosby MA, Card A, Liu J, Lindstrom WA, Chang DW. Immediate breast reconstruction and lymphedema incidence. Plast Reconst Sug 2012;129:789e-95e.
57. Fu MR, Axelrod D, Guth A, Fletcher J, Qiu JM, et al. Patterns of obesity and lymph fluid level during the first year of breast cancer treatment: a prospective study. J Pers Med 2015;5:326-40.
58. Petrek JA, Senie RT, Peters M, Rosen PP. Lymphedema in a cohort of breast carcinoma survivors 20 years after diagnosis. Cancer 2001;92:1368-77.
59. Helyer LK, Varnic M, Le LW, Leong W, McCready D. Obesity is a risk factor for developing postoperative lymphedema in breast cancer patients. Breast J 2010;16:48-54.
60. Ridner SH, Dietrich MS, Stewart BR, Armer JM. Body mass index and breast cancer treatment-related lymphedema. Support Care Cancer 2011;19:853-7.
61. Vignes S, Arrault M, Dupuy A. Factors associated with increased breast cancer-related lymphedema volume. Acta Oncol 2007;46:1138-42.
62. Jammallo LS, Miller CL, Singer M, Horick NK, Skolny MN, et al. Impact of body mass index and weight fluctuation on lymphedema risk in patients treated for breast cancer. Breast Cancer Res Treat 2013;142:59-67.
64. Smahel J. Adipose tissue in plastic surgery. Ann Plast Surg 1998;16:444-53.
65. Gaffney RM, Casley-Smith JR. Excess plasma proteins as a cause of chronic inflammation and lymphoedema: biochemical estimations. J Pathol 1981;133:229-42.
66. Executive Committee. The diagnosis and treatment of peripheral lymphedema: consensus document of the International Society of Lymphology. Lymphology 2016;49:170-84.
67. Campisi C, Boccardo F. Microsurgical techniques for lymphedema treatment: derivative lymphatic-venous microsurgery. World J Surg 2004;28:609-13.
68. Chang DW, Masia J, Garza R 3rd, Skoracki R, Neligan PC. Lymphedema: surgical and medical therapy. Plast Reconst Surg 2016;138:2095-185.
69. National Cancer Institute. Common Terminology Criteria for Adverse Events v4.0 (CTCAE). Available from: https://www.eortc.be/services/doc/ctc/CTCAE_4.03_2010-06-14_QuickReference_5x7.pdf [Last accessed on 23 Dec 2018].
70. Weissleder H, Weissleder R. Lymphedema: evaluation of qualitative and quantitative lymphoscintigraphy in 238 patients. Radiology 1998;167:729-35.
71. Witte CL, Witte MH, Unger EC, Williams WH, Bernas MJ, et al. Advances in imaging of lymph flow disorders. Radiographics 2000;20:1697-719.
72. Francis WP, Abghari P, Du W, Rymal C, Suna M, et al. Improving surgical outcomes: standardizing the reporting of incidence and severity of acute lymphedema after sentinel lymph node biopsy and axillary lymph node dissection. Am J Surg 2006;192:636-9.
73. Ridner SH, Montgomery LD, Hepworth JT, Stewart BR, Armer JM. Comparison of upper limb volume measurement techniques and arm symptoms between healthy volunteers and individuals with known lymphedema. Lymphology 2007;40:35-46.
74. Kyle UG, Bosaeus I, De Lorenzo AD, Deurenberg P, Elia M, et al. Bioelectrical impedance analysis--part I: review of principles and methods. Clinical Nutrition 2004;23:1226-43.
75. Jain MS, Danoff JV, Paul SM. Correlation between bioelectrical spectroscopy and perometry in assessment of upper extremity swelling. Lymphology 2010;43:85-94.
76. Warren AG, Janz BA, Slavin SA, Borud LJ. The use of bioimpedance analysis to evaluate lymphedema. Ann Plast Surg 2007;58:541-3.
77. Seward C, Skolny M, Brunelle C, Asdourian M, Salama L, et al. A comprehensive review of bioimpedance spectroscopy as a diagnostic tool for the detection and measurement of breast cancer-related lymphedema. J Surg Oncol 2016;114:537-42.
78. Shah C, Arthur DW, Wazer D, Khan A, Ridner S, et al. The impact of early detection and intervention of breast cancer-related lymphedema: a systemic review. Cancer Med 2016;5:1154-62.
79. Bicego D, Brown K, Ruddick M, Storey D, Wong C, et al. Exercise for women with or at risk for breast cancer-related lymphedema. Phys Ther 2006;86:1398-405.
80. Petrek JA, Pressman PI, Smith RA. Lymphedema: current issues in research and management. CA Cancer J Clin 2000;50:292-307.
81. National Cancer Institute. Lymphedema (PDQ): health professional version. Available from: https://www.cancer.gov/about-cancer/treatment/side-effects/lymphedema/lymphedema-hp-pdq [Last accessed on 26 Dec 2018].
82. Sayegh HE, Asdourian MS, Swaroop MN, Brunelle CL, Skolny MN, et al. Diagnostic methods, risk factors, prevention, and management of breast cancer-related lymphedema: past, present, and future directions. Curr Breast Cancer Rep 2017;9:111-21.
83. Ancukiewicz M, Miller CL, Skolny MN, O’Toole J, Warren LE, et al. Comparison of relative versus absolute arm size change as criteria for quantifying breast-cancer related lymphedema: the flaws in current studies and need for universal methodology. Breast Cancer Res Treat 2012;135:145-52.
84. Ancukiewicz M, Russell TA, O’Toole J, Specht M, Singer M, et al. A Standardized method for quantification of developing lymphedema in patients treated for breast cancer. Int J Radiat Oncol Biol Phys 2011;79:1436-43.
85. Miller CL, Specht MC, Horick N, Skolny MN, Jammallo LS, et al. A novel, validated method to quantify breast cancer-related lymphedema (BCRL) following bilateral breast surgery. Lymphology 2013;46:64-74.
86. Specht MC, Miller CL, Russell TA, Horick N, Skolny MN, et al. Defining a threshold for intervention in breast cancer-related lymphedema: what level of arm volume increase predicts progression? Breast Cancer Res Treat 2013;140:485-94.
87. Monnin-Delhom ED, Gallix BP, Achard C, Briel JM, Janbon C. High resolution unenhanced computed tomography in patients with swollen legs. Lymphology 2002;35:121-8.
88. Hudson TM, Hamlin DJ, Enneking WF, Pettersson H. Magnetic resonance imaging of bone and soft tissue tumors: early experience in 31 patients compared with computed tomography. Skeletal Radiol 1985;13:134-46.
89. Case TC, Witte CL, Witte MH, Unger EC, Williams WH. Magnetic resonance imaging in human lymphedema: comparison with lymphoscintigraphy. Magn Reson Imaging 1992;10:549-58.
90. Dimakakos PB, Stefanopolous T, Antoniades P, Antoniou A, Gouliamos A, et al. MRI and ultrasonographic findings in the investigation of lymphedema and lipedema. Int Surg 1997;82:411-6.
91. Astrom KG, Abdsaleh S, Brenning GC, Ahlstrom KH. MR imaging of primary, secondary, and mixed forms of lymphedema. Acta Radiol 2001;42:409-16.
92. Struk S, Honart JF, Qassemyar Q, Leymarie N, Sarfati B, et al. Use of indocyanine green angioagraphy in oncological and reconstructive breast surgery. An Chir Plast Esthet 2018;63:54-61.
93. Burnier P, Niddam J, Bosc R, Hersant B, Meningaud JP. Indocyanine green applications in plastic surgery: a review of the literature. J Plast Reconstr Aesthet Surg 2017;70:814-27.
94. Sorrentino L, Sartani A, Pietropaolo G, Bossi D, Mazzucchelli S, et al. A novel indocyanine green fluorescence-guided video-assisted technique for sentinel node biopsy in breast cancer. World J Surg 2018;42:2815-24.
95. Sugie T, Ikeda T, Kawaguchi A, Shimizu A, Toi M. Sentinel lymph node biopsy using indocynanine green fluorescence in early-stage breast cancer: a meta-analysis. Int J Clin Oncol 2017;22:11-17.
96. Garza RM, Ooi ASH, Falk J, Chang DW. The relationship between clinical and indocyanine green staging in lymphedema. Lymphat Res Biol 2018;18.
97. Mihara M, Hara H, Araki J, Kikuchi K, Narushima M, et al. Indocyanine green (ICG) lymphography is superior to lymphoscintigraphy for diagnostic imaging of early lymphedema of the upper limbs. PLoS One 2012;7:e38182.
98. Narushima M, Yamamoto T, Ogata F, Yoshimatsu H, Mihara M, et al. Indocyanine green lymphography findings in limb lymphedema. J Reconstr Microsurg 2016;32:72-9.
99. Badger CM, Peacock JL, Mortimer PS. A randomized, controlled, parallel-group clinical trial comparing multilayer bandaging followed by hosiery versus hosiery alone in the treatment of patients with lymphedema of the limb. Cancer 2000;88:2832-7.
101. Brorson H, Svensson H. Liposuction combined with controlled compression therapy alone. Plast Reconstr Surg 1998;102:1058-67.
102. Shao Y, Qi K, Zhou QH, Zhong DS. Intermittent pneumatic compression pump for breast cancer-related lymphedema: a sys¬tematic review and meta-analysis of randomized controlled trials. Oncol Res Treat 2014;37:170-4.
104. Vodder E. Lymph drainage ad modem Voder. Aesthet Med 1965;14:190-203.
105. McNeely ML, Magee DJ, Lees AW, Bagnall KM, Haykowsky M, et al. The addition of manual lymph drainage to compression therapy for breast cancer related lymphedema: a randomized controlled trial. Breast Cancer Res Treat 2004;86:95-106.
106. Andersen L, Hojris I, Erlandsen M, Andersen J. Treatment of breast cancer-related lymphedema with without manual lymphatic drainage: a randomized study. Acta Oncol 2000;39:399-405.
107. Didem K, Ufuk YS, Serdar S, Zumre A. The comparison of two different physiotherapy methods in treatment of lymphedema after breast surgery. Breast Cancer Res Treat 2005;93:49-54.
108. Lasinski BB. Complete decongestive therapy for treatment of lymphedema. Semin Oncol Nurs 2013;29:20-7.
109. Javid SH, Anderson BO. Mounting evidence against complex decongestive therapy as a first-line treatment for early lymphedema. J Clin Oncol 2013;31:3737-8.
110. Dayes IS, Whelan TJ, Julian JA, Parpia S, Pritchard KI, et al. Randomized trial of decon¬gestive lymphatic therapy for the treatment of lymphedema in women with breast cancer. J Clin Oncol 2013;31:3758-63.
111. Tidhar D, Drouin J, Shimony A. Aqua lymphatic therapy in managing lower extremity lymphedema. J Support Oncol 2007;5:179-83.
112. Tidhar D, Katz-Leurer. Aqua lymphatic therapy in women who suffer from breast cancer treatment-related lymphedema: a randomized controlled study. Support Care Cancer 2010;18:383-92.
113. Yeung W, Semciw AI. Aquatic therapy for people with lymphedema: a systemic review and meta-analysis. Lymphat Res Biol 2018;16:9-19.
114. Piller NB. Conservative treatment of acute and chronic lymph¬edema with benzopyrones. Lymphology 1976;9:132-7.
115. Casley-Smith JR, Morgan R, Piller NB. Treatment of lymphedema of the arms and legs with 5,6-benzo-[alpha]-pyrone. N Engl J Med 1993;329:1158-63.
116. Casley-Smith JR. Modern treatment of lymphoedema. II. The ben¬opyrones. Australas J Dermatol 1992;33:69-74.
117. Badger C, Preston N, Seers K, Mortimer P. Benzo-pyrones for reducing and controlling lymphedema of the limbs. Cochrane Database Syst Rev 2004;CD003140.
118. Charles H. Elephantiasis of the leg. In: Latham A, English T, editors. A system of treatment. London: Churchill; 1912.
119. Campisi C, Bellini C, Campisi C, Accogli S, Bonioli E, et al. Microsurgery for lymph¬edema: clinical research and long-term results. Microsurgery 2010;30:256-60.
120. Campisi CC, Ryan M, Boccardo F, Campisi C. A single-site tech¬nique of multiple lymphatic-venous anastomoses for the treatment of peripheral lymphedema: long-term clinical outcome. J Reconstr Microsurg 2016;32:42-9.
121. Weiss M, Baumeister RG, Hahn K. Post-therapeutic lymphedema: scintigraphy before and after autologous lymph vessel transplantation: 8 years of long-term follow-up. Clin Nucl Med 2002;27:788-92.
122. Hou C, Wu X, Jin X. Autologous bone marrow stomal cells trans¬plantation for the treatment of secondary arm lymphedema: a prospective controlled study in patients with breast cancer related lymphedema. Jpn J Clin Oncol 2008;38:670-4.
123. Gharb BB, Rampazzo A, Spanio di Spilimbergo S, Xu ES, Chung KP, et al. Vas¬cularized lymph node transfer based on the hilar perforators improves the outcome in upper limb lymphedema. Ann Plast Surg 2011;67:589-93.
124. Saaristo AM, Niemi TS, Viitanen TP, Tervala TV, Hartiala P, et al. Microvascular breast reconstruction and lymph node transfer for postmastectomy lymphedema patients. Ann Surg 2012;255:468-73.
125. Becker C, Assouad J, Riquet M, Hidden G. Postmastectomy lymphedema: long-term results following microsurgical lymph node transplantation. Ann Surg 2006;243:313-5.
126. Lin CH, Ali R, Chen SC, Wallace C, Chang YC, et al. Vascularized groin lymph node transfer using the wrist as a recipient site for management of post-mastectomy upper extremity lymphedema. Plast Reconstr Surg 2009;123:1265-75.
127. Dancey A, Nassimizadeh A, Nassimizadeh M, Warner RM, Waters R. A chimeric vascularized groin lymph node flap and DIEP flap for the management of lymphedema secondary to breast cancer. J Plast Reconstr Aesthet Surg 2013;66:735-7.
128. Patel KM, Manrique O, Sosin M, Hashmi MA, Poysophon P, et al. Lymphatic mapping and lymphedema surgery in the breast cancer patient. Gland Surg 2015;4:244-56.
130. Qi F, Gu J, Shi Y, Yang Y. Treatment of upper limb lymphedema with combination of liposuction, myocutaneous flap transfer and lymph-fascia grafting: a preliminary study. Microsurgery 2009;29:29-34.
131. Damstra RJ, Voesten HG, Klinkert P, Brorson H. Circumferential suction-assisted lipectomy for lymphoedema after surgery for breast cancer. Br J Surg 2009;96:859-64.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Ayre K, Parker C. Lymphedema after treatment of breast cancer: a comprehensive review. J Unexplored Med Data 2019;4:5. http://dx.doi.org/10.20517/2572-8180.2019.02
AMA Style
Ayre K, Parker C. Lymphedema after treatment of breast cancer: a comprehensive review. Journal of Unexplored Medical Data. 2019; 4: 5. http://dx.doi.org/10.20517/2572-8180.2019.02
Chicago/Turabian Style
Ayre, Kareen, Catherine Parker. 2019. "Lymphedema after treatment of breast cancer: a comprehensive review" Journal of Unexplored Medical Data. 4: 5. http://dx.doi.org/10.20517/2572-8180.2019.02
ACS Style
Ayre, K.; Parker C. Lymphedema after treatment of breast cancer: a comprehensive review. J. Unexplored. Med. Data. 2019, 4, 5. http://dx.doi.org/10.20517/2572-8180.2019.02
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 680 clicks
Cite This Article 680 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.