Cytotoxic effects of tamoxifen in breast cancer cells
Abstract
Aim: To investigate the cytotoxic effects of tamoxifen on the breast cancer cell line (MCF7).
Methods: The cytotoxic effects of tamoxifen on MCF7 cells were investigated using caspase-9 activity and high content screening assays. Apoptosis mechanisms following tamoxifen treatment were also investigated.
Results: The most significant cytotoxic effect of tamoxifen in MCF7 cells was a half-maximal inhibitory concentration (IC50) of 4.506 µg/mL. A significant increase in caspase-9 activity was also observed when MCF7 cells were treated with tamoxifen (5 µg/mL). Furthermore, increased cell membrane permeability, cytochrome c level, and nuclear intensity were observed with tamoxifen (100 µg/mL) compared with doxorubicin (20 µg/mL) treatment. However, a noticeable decrease in cell viability and mitochondrial membrane permeability was observed with tamoxifen (100 µg/mL) treatment compared with doxorubicin (20 µg/mL) as a positive control.
Conclusion: Tamoxifen showed in vitro cytotoxic effects in MCF7 cells as demonstrated by high-content screening and caspase-9 activity assays. Tamoxifen inhibits estrogen mechanisms, although toxic effect was observed.
Keywords
Introduction
Cancer is a common cause of death nationwide[1], with the number of new cases of cancer in the US was estimated to be ca. 1.7 million in 2016[2]. Although the number of cancer deaths was estimated at more than half a million in the US in 2016, this number has dropped by ca. 23% since 1991[2]. Health care for cancer patients requires extensive financial resources. For example, the health care costs for cancer patients in the US were ca. $156 billion in 2010, and are expected to increase further to $156 billion by 2020[3]. Breast cancer is projected to be among the most common cancer[3], and is the most frequent malignant neoplasm in women[4].
Metastasis is the main cause of death from breast cancer and is responsible for 90% of total breast cancer deaths[5,6]. Although progress has been made in cancer treatment and techniques used to detect its progression, breast cancer continues to have a high mortality among women[7,8]. Radiation, chemotherapy, and surgery can be used to control tumor growth, but these options need to be effective to manage breast cancer metastases[9-12]. Protease enzymes such as caspases exhibit cysteine protease activity and play an important role in apoptosis or programmed cell death[13,14].
Caspases are capable of cleaving various proteins in different cells[15]. Such processes can control the activation and/or inactivation of other proteins. In addition, caspases participate in nuclear fragmentation, chromatin condensation, creation of active signaling molecules, cell contraction, and other biochemical changes associated with apoptosis[15-17]. Various types of caspases are classified primarily based on their roles in apoptosis and mode of action[16]. For example, caspase-8 and -9 are known as initiator caspases, while caspase-3, -6, and -7 are known as executioner caspases[16]. Progress has been made in the early diagnosis and treatment of cancer, and in particular, breast cancer[18-22]. The MCF7 cancer cell line can be used to sensitively detect the response of estrogen[23,24]. We have previously investigated the cytotoxicity and anticancer activities of several aryl phosphonates on MCF7 cells[25]. Recently, we also reported the cytotoxic activities of anastrozole in several cancer cell lines[26,27]. In the current study, we report the cytotoxic effects of tamoxifen in MCF7 cancer cells.
Methods
Data
Data were collected using Excel (Microsoft Office 2010, Microsoft Corp., Redmond, WA). SPSS software (IBM Software, version 22) was used to analyze the data.
Cell culture
The MCF7 cancer cell line was purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). Cells were cultured in Dulbecco’s Modified Eagle medium (Life Technologies, Inc., Rockville, MD, USA) supplemented with heat-inactivated fetal bovine serum (10%; Sigma-Aldrich, St. Louis, MO, USA), streptomycin, penicillin (1%), and glutamine (2 mmol/L).
Biological activity
Methylthiazol tetrazolium (MTT) assays were carried out at the Biotechnology Research Centre, Al-Nahrain University, Baghdad, Iraq between October 2016 and February 2017. Caspase-9 and high content screening (HCS) assays were carried out at the Natural Product Research and Drug Discovery Centre, Department of Pharmacology, Faculty of Medicine, University of Malaya, Kuala Lumpur, Malaysia between October 2016 and February 2017. The MTT and HCS assays were performed as described previously[28].
Cytotoxicity assay (MTT assay)
The MTT assay was carried out in the MCF7 cell line to determine the anticancer activity of tamoxifen. Tamoxifen was dissolved in dimethyl sulfoxide (DMSO) to produce a stock solution and serial dilutions were prepared (0.78125-200 μg/mL). Tamoxifen (100 μL) and doxorubicin, as a control, were added to MCF7 cells and the cell cultures were incubated for 24 h in a CO2 incubator. MTT (5 μg/mL) was added to each well and the plates were incubated further for 1-4 h. The media was removed and DMSO was added to each well to solubilize the formazan crystals. The absorbance was measured by the use of a Hidex Chameleon microplate reader (LabLogic Systems Ltd., Sheffield, United Kingdom) at 575 nm.
High-content screening
A Thermo Scientific Cellomics multi-parameter cytotoxicity 3 kit (Thermo Scientific, Japan) was used for the simultaneous detection of cell viability, nuclear intensity, cell membrane permeability, mitochondrial membrane potential, and cytochrome c level in the MCF7 cell line. The kit contained cytochrome c as a primary antibody, DyLight™ 649 conjugated goat anti-mouse IgG, Hoechst dyes, wash buffer (10× Dulbecco’s phosphate buffered saline [PBS]), permeabilization buffer (10× Dulbecco’s PBS with 1% Triton® X-100), and a blocking buffer (10×). The distribution and intensity of fluorescence within cells were imaged (n = 5) using an HCS system (Thermo Scientific, Japan). The system was attached to a computerized imaging microscope equipped with a Zeiss 40× (0.75 NA) Plan-Neofluar objective lens. Cells were treated with tamoxifen for 24 h followed by the addition of mitochondrial membrane potential (MMP) and the cell permeability dyes and incubated for 30 min at 37 °C. Cells were fixed and permeabilized using a standard procedure[28].
Hoechst 33342 staining assay
MCF7 cells were cultured in 6-well plates for 24 h. Changes in MCF7 cell nuclear morphology were observed using a Zeiss Axio Observer microscope (Thermo Scientific, Japan).
Results
Cytotoxic effects of tamoxifen on MCF7 cell viability
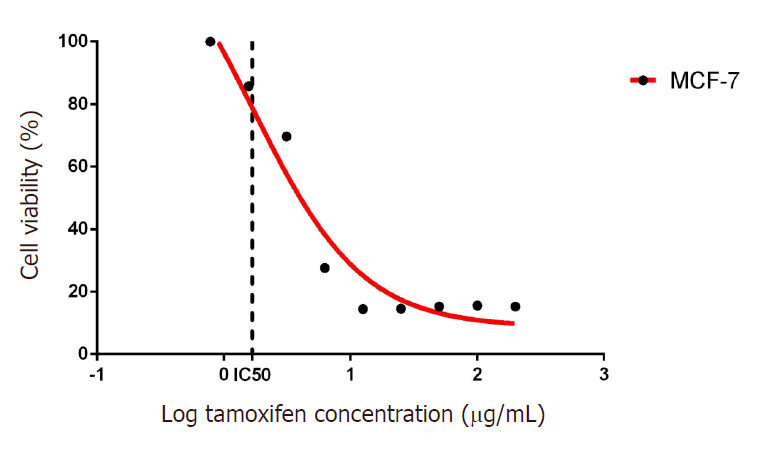
The cytotoxic effects of tamoxifen were measured in the MCF7 cell line using the MTT method[26]. MCF7 cells were treated with various concentrations of tamoxifen (200, 100, 50, 25, 12.5, 6.25, 3.125, 1.5625, and 0.78125 μg/mL) for 24 h at 37 °C. The viability rate of MCF7 cells treated with tamoxifen is shown in Figure 1, and it was found to be 15.28%, 15.60%, 15.29%, 14.58%, 14.42%, 27.65%, 69.65%, 85.73%, and 100%, respectively. Tamoxifen cytotoxicity thus increased with increasing concentration, and the highest cytotoxic activity (15.60%) was observed at a concentration of 100 µg/mL with an IC50 of 4.506 µg/mL.
Effects of tamoxifen on MCF7 cell caspase-9 activity
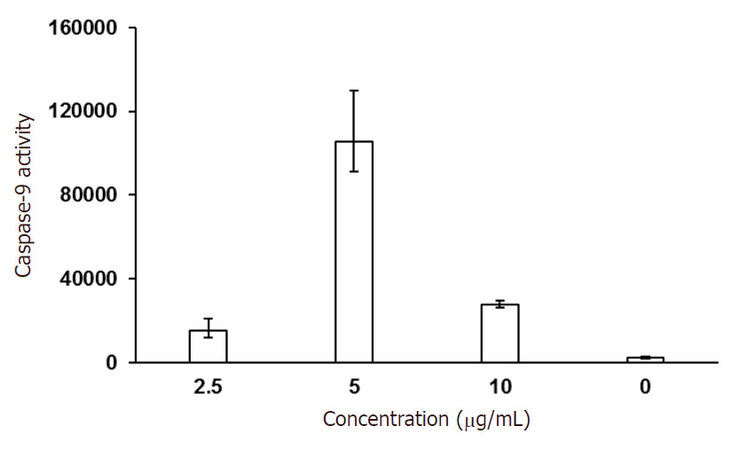
A significant increase in the mean activity of caspase-9 (105,428 ± 24,628) was observed in MCF7 cells treated with tamoxifen (5 µg/mL), at an IC50 of 4.506 µg/mL [Figure 2].
Cytotoxic effects of tamoxifen on MCF7 cells using HCS
The cytotoxicity of tamoxifen in MCF7 cells over 24 h was evaluated by HCS. Five tamoxifen concentrations (100, 50, 25, 12.5, and 6.25 µg/mL) were used to detect changes in MCF7 cell viability, nuclear intensity, membrane permeability, MMP, and cytochrome c. Table 1 shows the changes in these 5 parameters in addition to the least significant differences (LSD). The highest significant changes in valid cell count, nuclear intensity, MMP, and cytochrome c were observed at a tamoxifen concentration of 100 µg/mL, compared with doxorubicin (20 µmol/L) as a positive control (P < 0.01). Lower concentrations of tamoxifen (12.5 and 6.25 µg/mL) did not cause noticeable changes in the evaluated parameters and results were similar to those obtained for untreated cells (negative control).
Cytotoxic effects of tamoxifen on cellular parameters by HCS
| Concentration (μg/mL) | HCS parameter (mean ± SD) | ||||
|---|---|---|---|---|---|
| CV | NI | CP | MMP | CC | |
| Untreated cells (0) | 1279 ± 5.657a | 402.0 ± 16.97d | 72.00 ± 8.485c,d | 384.0 ± 41.01b | 212.5 ± 31.82d |
| 6.25 | 1073 ± 66.47b | 418.5 ± 3.536d | 85.50 ± 7.778c | 376.5 ± 47.38b | 211.5 ± 26.16d |
| 12.5 | 1039 ± 86.97b | 422.5 ± 3.536d | 70.00 ± 18.38c,d | 416.0 ± 18.38a | 225.0 ± 14.14d |
| 25 | 940.5 ± 16.26b,c | 407.5 ± 10.61d | 61.50 ± 21.92d | 418.5 ± 16.26a | 191.0 ± 36.77d |
| 50 | 974.0 ± 73.54b,c | 504.5 ± 7.778c | 86.00 ± 4.243c | 299.0 ± 11.31c | 283.5 ± 23.33c |
| 100 | 854.5 ± 181.7c | 661.5 ± 67.18b | 112.0 ± 12.73b | 174.0 ± 24.04d | 421.0 ± 55.15b |
| Doxorubicin | 519.5 ± 89.80d | 951.5 ± 54.45a | 187.5 ± 17.68a | 99.00 ± 5.657e | 650.0 ± 70.71a |
| LSD value* | 178.371 | 48.227 | 22.194 | 46.842 | 48.053 |
Cell viability
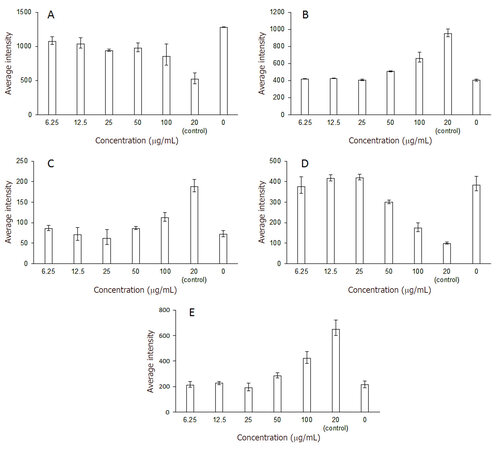
As shown in Figure 3A, a significant reduction in MCF7 cell viability was observed with increasing tamoxifen concentration over 24 h. The percentage cell viability in cells treated with tamoxifen was 33.19% (100 µg/mL), 23.8% (50 µg/mL), 26.46% (25 µg/mL), 18.76% (12.5 µg/mL), and 16.11% (6.25 µg/mL). The differences between the results obtained for the positive control and those for all other experimental groups were statistically significant. The most significant reduction (P = 0.0101) in cell count (854.5) compared with doxorubicin (519.5) was observed with 100 µg/mL tamoxifen (P = 0.0003).
Nuclear intensity
MCF7 cell nuclear intensity [Figure 3B] was significantly increased (64.55%; P = 0.0005) with 100 µg/mL tamoxifen compared with doxorubicin (20 μg/mL) as a positive control. Nuclear intensities following treatment with other tamoxifen concentrations (50-6.25 µg/mL) showed no significant difference (25.49%-4.10%) compared with the control (P < 0.0001). Similar results were previously obtained following treatment of MCF7 cells with anastrozole[27].
Cell membrane permeability
MCF7 cell membrane permeability [Figure 3C] was significantly increased (55.56%; P = 0.0072) following treatment with 100 µg/mL tamoxifen, compared with doxorubicin (20 μg/mL). MCF7 cell membrane permeability at other tamoxifen concentrations (50-6.25 µg/mL) showed no significant difference (25.49%-4.10%) compared with control (P = 0.0004).
Mitochondrial membrane permeability
MCF7 MMP intensity increased with increasing tamoxifen concentration [Figure 3D]. The highest MMP intensity (54.68%) was detected at tamoxifen dose of 100 µg/mL compared to that of the standard (P < 0.0001).
Cytochrome c
Treatment of MCF7 cells with tamoxifen led to a dose-dependent increase in the intensity of cytochrome c released [Figure 3E]. At a dose of 100 μg/mL of tamoxifen, cytochrome c intensity was 64.80% compared with the intensity obtained with the control (20 μg/mL doxorubicin). There were no significant differences between the different concentrations of tamoxifen, and all values were significantly lower than the positive control (P < 0.0001).
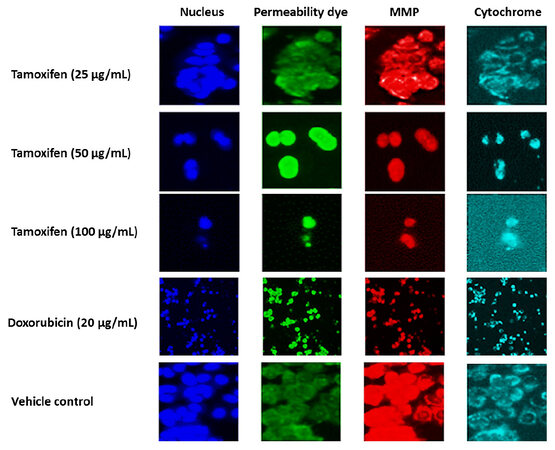
Images of MCF7 cells treated with tamoxifen (100 μg/mL), doxorubicin (20 μg/mL) as a positive control, and culture medium as a negative control for 24 h at 37 °C are shown in Figure 4. Cells were stained with Hoechst 33342 dye (excitation 330/emission 420), which enables monitoring of cell loss, cell membrane permeability dye (excitation 491/emission 509), MMP dye (excitation 552/emission 576) for mitochondrial membrane potential changes, and goat anti-mouse secondary antibody conjugated with DyLightTM 649 for cytochrome c release. Tamoxifen (100 μg/mL) stained the MCF7 cells, and the level of staining was similar to that of doxorubicin (20 μg/mL).
Discussion
Caspase-9 activity directly correlates with the toxic effects of tamoxifen. Tamoxifen can affect the cell membranes, causing rapid changes in membrane permeability. Such effects can lead to cell death and a reduction in cell viability[29]. In addition, tamoxifen rapidly inhibits estrogen-dependent protein kinase C in MCF7 cells[30] and induces rapid mitochondrial death in estrogen receptor-positive MCF7 cells[31]. In this study, a significant decrease in the mean caspase-9 activity was observed when MCF7 cells were treated with tamoxifen (10 µg/mL). Tamoxifen is a potent antagonist of estrogen and induces apoptosis in estrogen receptor-positive cells. However, at concentrations higher than 5 μg/mL, it shows estrogenic behavior and acts as an agonist of estrogen[32]. The tamoxifen-induced activation of the intrinsic caspase pathway in breast cancer cells was statistically significant (P < 0.0001 at 5 µg/mL). Compelling evidence has shown that the majority of cytotoxic drugs initiate apoptosis by triggering the cytochrome c/Apaf-1/caspase-9-dependent pathway through the mitochondrion[33].
Cell viability is an important toxicity assay parameter and is directly associated with the toxic effects of a drug[34]. Tamoxifen induced a reduction in cell viability in MCF7 cells. The reduction was found to be dependent on cell density and tamoxifen concentration. Such effects may be attributed to cytostatic and/or cytocide effects that might down-regulate telomerase activity[35].
Changes in cell membrane permeability are often associated with a toxic or apoptotic response[36]. In addition, cytotoxicity can lead to the loss of cell membrane integrity[36]. Tamoxifen can alter the composition and physical order of lipids within intracellular and plasma membranes. Such effects lead to a change in plasma membrane permeability due to second messenger formation through the phospholipase pathway and the sustained activation of protein kinase C[37]. The increased plasma membrane permeability, along with the sustained protein kinase C activity, can affect the binding of rod outer segments[37].
The intensity of MCF7 mitochondrial membrane permeability increased with increasing tamoxifen concentration, possibly because cells underwent either necrosis or apoptosis accompanied by changes in mitochondrial function. Such changes could lead to the loss of mitochondrial membrane potential and the release of cytochrome c from the mitochondria[38].
Cytochrome c intensity also increased with increasing tamoxifen concentration. Caspase-9 activation may lead to caspase-3 formation, which in turn causes apoptosis and increases extracellular signal-regulated kinase (ERK) phosphorylation[39]. The findings of the present study suggest that tamoxifen has cytotoxic effects in MCF7 cells. Cytotoxicity was apparent at 100 µg/mL of tamoxifen, with a 33.19% cell loss and a 64.55% change in the nuclear morphology.
Cytochrome c plays an important role in apoptosis and can be released into the cytosol from the mitochondria[40]. MCF7 cells treated with tamoxifen showed strong nuclear staining for cytochrome c compared with doxorubicin [Figure 4]. Clearly, the use of tamoxifen to treat breast cancer cells can imitate the cytochrome c from the mitochondria to the cytosol.
Figure 4. Multiparameter cytotoxicity analysis of MCF7 cells treated with tamoxifen. MCF7: breast cancer cell line; MMP: mitochondrial membrane potential
In conclusion, tamoxifen exhibits cytotoxic effects in MCF7 breast cancer cells. The effect of tamoxifen on apoptosis was shown as an increase in caspase-9 activity at a concentration of 5 µg/mL and IC50 of 4.506 µg/mL. The HCS assay showed that tamoxifen has toxic effects in MCF7 cells at 100 μg/mL in a dose-dependent manner, with increased cell nuclear intensity, membrane permeability, and cytochrome c observed. Furthermore, cell viability was decreased, along with a change in mitochondrial membrane potential.
Declarations
Authors’ contributionsConceived and designed the experiments, discussed the results and improved the final text of the paper: Hassan F, Mohammed G, El-Hiti GA, Alshanon A, Yousif E
Data source and availabilityPresentation in the paper of the data/materials/code accurately reflects the original sources.
Financial support and sponsorshipThe project was supported by by Al-Nahrain University and King Saud University.
Conflicts of interestThere are no conflicts of interest.
Patient consentNot applicable.
Ethics approvalThis study was approved by our review board.
Copyright© The Author(s) 2018.
REFERENCES
1. Van Hemelrijck M, Folkvaljon Y, Adolfsson J, Akre O, Holmberg L, Garmo H, Stattin P. Causes of death in men with localized prostate cancer: a nationwide, population-based study. BJU Int 2016;117:507-14.
3. NIH National Cancer Institute. Cancer Statistics. Available from: https://www.cancer.gov/about-cancer/understanding/statistics. [Last accessed on 6 Feb 2018].
4. Patsialou A, Wang Y, Lin J, Whitney K, Goswami S, Kenny PA, Condeelis JS. Selective gene-expression profiling of migratory tumor cells in vivo predicts clinical outcome in breast cancer patients. Breast Cancer Res 2012;14:R139.
5. Kozłowski J, Kozłowska A, Kocki J. Breast cancer metastasis-insight into selected molecular mechanisms of the phenomenon. Postepy Hig Med Dosw (Online) 2015;69:447-51.
6. Spano D, Heck C, De Antonellis P, Christofori G, Zollo M. Molecular networks that regulate cancer metastasis. Semin Cancer Biol 2012;22:234-49.
7. Youlden DR, Cramb SM, Yip CH, Baade PD. Incidence and mortality of female breast cancer in the Asia-Pacific region. Cancer Biol Med 2014;11:101-15.
8. Lorusso G, Rüegg C. New insights into the mechanisms of organ-specific breast cancer metastasis. Semin Cancer Biol 2012;22:226-33.
9. Ghoncheh M, Pournamdar Z, Salehiniya H. Incidence and mortality and epidemiology of breast cancer in the world. Asian Pac J Cancer Prev 2016;17:43-6.
10. Nounou MI, ElAmrawy F, Ahmed N, Abdelraouf K, Goda S, Syed-Sha-Qhattal H. Breast cancer: conventional diagnosis and treatment modalities and recent patents and technologies. Breast Cancer (Auckl) 2015;9:17-34.
11. Pillai G, Ceballos-Coronel ML. Science and technology of the emerging nanomedicines in cancer therapy: a primer for physicians and pharmacists. SAGE Open Med 2013;1:2050312113513759.
12. Brown S, Khan DR. The treatment of breast cancer using liposome technology. J Drug Deliv 2012;2012:212965.
13. Jelínek M, Balušíková K, Schmiedlová M, Němcová-Fürstová V, Šrámek J, Stančíková J, Zanardi I, Ojima I, Kovář J. The role of individual caspases in cell death induction by taxanes in breast cancer cells. Cancer Cell Int 2015;15:8.
14. Chávez-Mardones J, Gallardo-Escárate C. Immune response of apoptosis-related cysteine peptidases from the red abalone Haliotis rufescens (HrCas8 and HrCas3): molecular characterization and transcription expression. Fish Shellfish Immunol 2014;39:90-8.
15. Denning DP, Hatch V, Horvitz HR. Both the caspase CSP-1 and a caspases-independent pathway promote programmed cell death in parallel to the canonical pathway for apoptosis in Caenorhabditis elegans. PLoS Genet 2013;9:e1003341.
16. McIlwain DR, Berger T, Mak TW. Caspase functions in cell death and disease. Cold Spring Harb Perspect Biol 2013;5:a008656.
17. Würstle ML, Laussmann MA, Rehm M. The central role of initiator caspase-9 in apoptosis signal transduction and the regulation of its activation and activity on the apoptosome. Exp Cell Res 2012;318:1213-20.
18. Balekouzou A, Yin P, Pamatika CM, Bekolo CE, Nambei SW, Djeintote M, Kota K, Mossoro-Kpinde CD, Shu C, Yin M, Fu Z, Qing T, Yan M, Zhang J, Chen S, Li H, Xu Z, Koffi B. Reproductive risk factors associated with breast cancer in women in Bangui: a case-control study. BMC Womens Health 2017;17:14.
19. Khan MSS, Salam MA, Haque RSMA, Abdul Majid AMS, Bin Abdul Majid AS, Asif M, Basheer KA, Tabana YM. Synthesis, cytotoxicity, and long-term single dose anti-cancer pharmacological evaluation of dimethyltin(IV) complex of N(4)-methylthiosemicarbazone (having ONS donor ligand). Cogent Biol 2016;2:1154282.
20. Attia YM, El-Abhar HS, Al Marzabani MM, Shouman SA. Targeting glycolysis by 3-bromopyruvate improves tamoxifen cytotoxicity of breast cancer cell lines. BMC Cancer 2015;15:838.
21. Xie X, Wu MY, Shou LM, Chen LP, Gong FR, Chen K, Li DM, Duan WM, Xie YF, Mao YX, Li W, Tao M. Tamoxifen enhances the anticancer effect of cantharidin and norcantharidin in pancreatic cancer cell lines through inhibition of the protein kinase C signaling pathway. Oncol Lett 2015;9:837-44.
22. Prakash O, Kumar A, Kumar P, Ajeet A. Anticancer potential of plants and natural products: a review. Am J Pharmacol Sci 2013;1:104-15.
23. Sweeney EE, McDaniel RE, Maximov PY, Fan P, Jordan VC. Models and mechanisms of acquired antihormone resistance in breast cancer: significant clinical progress despite limitations. Horm Mol Biol Clin Investig 2012;9:143-63.
24. Hole S, Pedersen AM, Hansen SK, Lundqvist J, Yde CW, Lykkesfeldt AE. New cell culture model for aromatase inhibitor-resistant breast cancer shows sensitivity to fulvestrant treatment and cross-resistance between letrozole and exemestane. Int J Oncol 2015;46:1481-90.
25. Abdel-Megeed MF, Badr BE, Azaam MM, El-Hiti GA. Synthesis, antimicrobial and anticancer activities of a novel series of diphenyl 1-(pyridin-3-yl)ethylphosphonates. Bioorg Med Chem 2012;20:2252-8.
26. Hassan F, Abdul-Hameed A, Alshanon A, Abdullah MB, Huri HZ, Hairunisa N, Yousif E. Antitumor activity for gold (III) complex by high content screening technique (HCS) and cell viability assay. Asian J Biochem 2015;10:252-66.
27. Hassan F, El-Hiti GA, Abd-Allateef M, Yousif E. Cytotoxicity anticancer activities of anastrozole against breast, liver hepatocellular, and prostate cancer cells. Saudi Med J 2017;38:359-65.
28. Duellman SJ, Zhou W, Meisenheimer P, Vidugiris G, Cali JJ, Gautam P, Wennerberg K, Vidugiriene J. Bioluminescent, nonlytic, real-time cell viability assay and use in inhibitor screening. Assay Drug Dev Tech 2015;13:456-65.
29. Mohammed SA, Hassan FK, Philip AK, Abd-Allateef M, Yousif E. Role of aromatase and anastrozole in cancer treatment. Int J Pharm Sci Rev Res 2016;40:135-40.
30. Boyan BD, Sylvia VL, Frambach T, Lohmann CH, Dietl J, Dean DD, Schwartz Z. Estrogen-dependent rapid activation of protein kinase C in estrogen receptor-positive MCF-7 breast cancer cells and estrogen receptor-negative HCC38 cells is membrane-mediated and inhibited by tamoxifen. Endocrinology 2003;144:1812-24.
31. Razandi M, Pedram A, Jordan VC, Fuqua S, Levin ER. Tamoxifen regulates cell fate through mitochondrial estrogen receptor beta in breast cancer. Oncogene 2013;32:3274-85.
32. Ma G, Pan Y, Zhou C, Sun R, Bai J, Liu P, Ren Y, He J. Mitogen-activated protein kinase phosphatase 1 is involved in tamoxifen resistance in MCF7 cells. Oncol Rep 2015;34:2423-30.
33. Zhou B, Blanchard A, Wang N, Ma X, Han J, Schroedter I, Leygue E, Myal Y. Claudin 1 promotes migration and increases sensitivity to tamoxifen and anticancer drugs in luminal-like human breast cancer cells MCF7. Cancer Invest 2015;33:429-39.
34. Al-Jailawi MH, Nasir HM, Aziz GM. Cytotoxic effect of biosurfactants produced by novel thermophillic Geobacillus thermoleovorans (JQ 912239). Int J Adv Res 2015;3:632-7.
35. Ajabnoor GM, Crook T, Coley HM. Paclitaxel resistance is associated with switch from apoptotic to autophagic cell death in MCF-7 breast cancer cells. Cell Death Dis 2012;3:e260.
36. Rodriguez Salgueiro S, González Núñez L, García Del Barco Herrera D, Santos Febles E, Maza Ares D, Millares López R, Berlanga Acosta J. Role of epidermal growth factor and growth hormone-releasing peptide-6 in acceleration of renal tissue repair after kanamycin overdosing in rats. Iran J Kidney Dis 2014;8:382-8.
37. Khadka NK, Cheng X, Ho CS, Katsaras J, Pan J. Interactions of the anticancer drug tamoxifen with lipid membranes. Biophys J 2015;108:2492-501.
38. Kim R, Emi M, Tanabe K. Role of mitochondria as the gardens of cell death. Cancer Chemother Pharmacol 2006;57:545-53.
39. Cha JD, Kim YH, Kim JY. Essential oil and 1,8-cineole from Artemisia lavandulaefolia induces apoptosis in KB cells via mitochondrial stress and caspase activation. Food Sci Biotechnol 2010;19:185-91.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Hassan F, Mohammed G, El-Hiti GA, Alshanon A, Yousif E. Cytotoxic effects of tamoxifen in breast cancer cells. J Unexplored Med Data 2018;3:3. http://dx.doi.org/10.20517/2572-8180.2017.25
AMA Style
Hassan F, Mohammed G, El-Hiti GA, Alshanon A, Yousif E. Cytotoxic effects of tamoxifen in breast cancer cells. Journal of Unexplored Medical Data. 2018; 3: 3. http://dx.doi.org/10.20517/2572-8180.2017.25
Chicago/Turabian Style
Hassan, Firas, Ghufran Mohammed, Gamal A. El-Hiti, Ahemd Alshanon, Emad Yousif. 2018. "Cytotoxic effects of tamoxifen in breast cancer cells" Journal of Unexplored Medical Data. 3: 3. http://dx.doi.org/10.20517/2572-8180.2017.25
ACS Style
Hassan, F.; Mohammed G.; El-Hiti GA.; Alshanon A.; Yousif E. Cytotoxic effects of tamoxifen in breast cancer cells. J. Unexplored. Med. Data. 2018, 3, 3. http://dx.doi.org/10.20517/2572-8180.2017.25
About This Article
Copyright
Data & Comments
Data
 Cite This Article 60 clicks
Cite This Article 60 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.