Surgical outcomes of distal splenorenal shunt or liver transplantation in treatment of schistosomal refractory variceal bleeding
Abstract
Schistosomiasis is one of the most prevalent neglected tropical diseases, remains a serious public health problem in many developing countries in the tropics and subtropics and its pathogenesis depends on the parasite-host interaction. Periportal fibrosis of schistosomiasis complicated by portal hypertension results from increased intrahepatic vascular resistance and blood flow through the portal venous system. This leads to portosystemic collateral variceal veins formation which dilates until they finally rupture and bleed due to progressive rising in the portal venous pressure. The clinical impact of esophagogastric varices is critical due to the rebleeding rate of esophageal varices remains high within two years after cessation of acute bleeding from medium or large varices. In the present review, we will discuss hepatosplenic schistosomiasis and oesophagogastric varices pathophysiology and review the current surgical outcomes of distal splenorenal shunt or liver transplantation in schistosomal refractory variceal bleeding.
Keywords
Introduction
Oesophagogastric varices are portosystemic collateral venous channels and their acute bleeding is a lethal complication of portal hypertension represented in 70% of cases[1]. Small varices dilated gradually at a rate of 5% per year and the risk of bleeding increased by variceal size, liver disease severity and red wale mark appearance[2]. The variceal rebleeding risk is highest within the first 6 weeks with a peak in the first 5 days[1]. In Sudan, a country endemic with schistosomiasis, with prevalence rates increased to 23.7% in 2012[3]. Portal hypertension due to schistosomal periportal fibrosis caused acute variceal bleeding in the majority of cases while liver cirrhosis is less common[4]. The mortality rate of schistosomiasis in published data from a village endemic with schistosomiasis in the Gezira state located in Sudan was 51/100,000 per year. The fatality rate per year is 1/1000 in patients secreting eggs and 11/100 in variceal bleeding patients[5]. Schistosoma mansoni (S.mansoni) complicated by portal hypertension has a major impact on morbidity and mortality because of esophagogastric variceal bleeding possibility[6]. Ten to twenty percent of patients were not controlled with endoscopic and pharmacological therapy, known as refractory variceal bleeding which needs more aggressive therapies to deal with it[7,8]. Although surgical procedures were less frequently performed with endoscopic therapy and transjugular intrahepatic portosystemic shunt (TIPS) development, it controls variceal rebleeding effectively. Furthermore, it indicated in patients with well-preserved liver function, treating hypersplenism and rarely inducing hepatic encephalopathy[9].
Hepatosplenic schistosomiasis overview
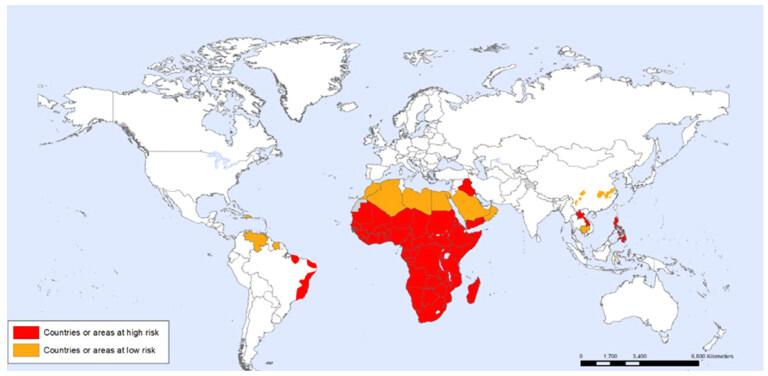
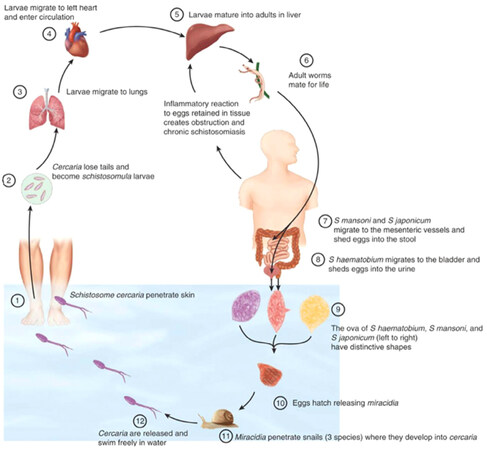
It’s a chronic disease caused by trematode blood fluke of genus Schistosoma-related to the Schistosomatidae family[10]. Sometimes called Bilharziasis or snail fever referred to Theodor Bilharz, a German surgeon working at Cairo Medical School who discovered the fluke in 1851 within the mesenteric veins of an Egyptian boy at autopsy [Figure 1]. Then described terminal spine mature ova and lateral spine immature ova, produced by the female worms[11,12]. Furthermore, McDonagh[13] has introduced antimony as an anti-bilharzial agent[14]. The World Health Organization includes schistosomiasis as a neglected tropical disease. The disease is believed to affect approximately 200 million individuals in many countries and kills more than 100,000 people per year, with most cases occurring in Africa, the Eastern Mediterranean and the Americas [Figure 2]. On an average 85% of these cases are indigenous to the African continent[12]. Furthermore, the disease is considered a risk for 650 million people living in endemic rural areas of tropical countries and associated with poor socioeconomic status[12,15]. The movement of people from endemic zones with unplanned dam establishment and irrigation projects led to newer endemic foci appearance. Thus, there is a need for an effective schistosomal control program including of health and hygiene education, reduction of water contamination through sanitary and safe water supply facilities, snail control measures and mass chemotherapy to eliminate the threat from several endemic countries[12]. In the endemic areas, children, women, fishermen, and farmers in irrigation channels are often infected with schistosomes and the pathogenic changes in schistosomiasis depend on interplay between host and parasite factors[12,16]. The life cycle characterized by the adult worms live in terminal venules of the bowel (S.mansoni) or bladder (Schistosoma haematobium) which passed their eggs through human feces or urine to reach fresh water. A larval form is released to infect snails (the intermediate host) to develop into cercariae which leave the snails, enter the water, and through the skin or mucous membranes penetration become schistosomula causing cercarial dermatitis or swimmer’s itch that may persist from few hours up to one week manifested by macular rash most commonly seen on the legs and feet[17,18]. Inside the human body, schistosomula, enter the vasculature of the host, then migrate to the lungs and remain there for several days before travelling to the liver. They feed on red blood cells which maturate and mate in liver vessels to form adult worms, living in the venous system where the females lay their eggs[19-21]. Most of the eggs carried upstream via the portal veins to the liver and trapped in the pre-sinusoidal portal venules[21]. Some eggs retained in the bowel or bladder wall while others passed into the lumen of intestine or bladder with feces or urine through their lytic secretions or carried proteases [Figure 3]. In advanced hepatosplenic schistosomiasis, the direct effect of ova deposition in the portal tributaries that it cause granuloma formation defined as inflammation surrounding eggs and limits adjacent tissues damage from its secreted antigens which replaced overtime (16-20 weeks post-infection) with periportal fibrosis[17]. At this stage, patients complain of abdominal pain, anorexia and diarrhea. Subsequently, hepatosplenomegaly and portal hypertension complicated by esophageal variceal bleeding, ascites and hepatic coma indicate the end stage of liver disease[20,22,23].
Diagnostic procedures include the demonstration of ova in stool which occurs after 45 days of infection sometimes has low sensitivity and if negative the rectal biopsy has a beneficial role in diagnosis and healing control[24,25]. Recently, the common liver injury indicators in hepatosplenic schistosomiasis are alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase and bilirubin levels which are higher twice than in healthy individuals, while gamma-glutamyl transferase (γ-GT) is five-fold higher linked to possible biliary tree anatomical changes caused by fibrosis in the portal region[26,27]. Hence, γ-GT levels are the best marker of progressive hepatic fibrosis and evaluation of liver dysfunction progression in schistosomiasis[28].
Patients with schistosomiasis have prolongation of the bleeding profile including prothrombin time, partial thromboplastin time, thrombin time and hypofibrinogenemia which are well-established findings[29]. Furthermore, these laboratory data are independently correlated with the presence of large esophageal varices or combined in the Child-Pugh score. The ultrasonography of the liver giving data on the liver size, echotexture, margins and degree of ascites[30]. Also, it assesses the underlying periportal fibrosis severity by measuring the portal tract thickness in which reflects the hemodynamic changes and provides a good estimate clinical status of patients with periportal fibrosis which graded from I-III as follow: Grade I include 3-5 mm echogenic thickening of the portal venous tract while Grade II is if the thickness between 5-7 mm with marked central lucency narrowing and Grade III if it’s more than 7 mm with central lucency obliteration and extension of the irregular thickening down to the main portal venous wall[31,32]. Sometimes it discovers the distention of portal and splenic veins exceeding 12 or 10 mm width respectively[25]. Furthermore, color doppler ultrasound showed hepatic left lobe hypertrophy and right lobe atrophy which was commonly seen in the hepatosplenic schistosomiasis[33].
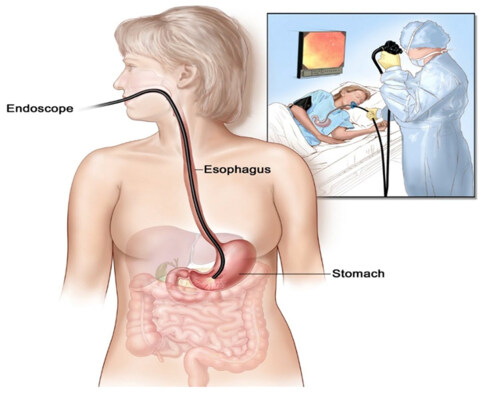
The oesophagogastroduodenoscopy (OGD) can demonstrate esophagogastric varices [Figure 4]. The liver biopsy is an invasive procedure not considered as the first diagnostic choice in which shows Symmer’s or pipe-stem fibrosis and the parenchyma between fibrotic areas is typically well-preserved their function. In patients with schistosomiasis alone the liver cirrhosis is rare but if co-infected with hepatitis B or C virus, progression to cirrhosis is brisk and this is common in endemic regions like Egypt and the Middle East[20,25,34-38]. Currently, schistosomal control was performed by praziquantel therapy with a single dose of 40 mg/kg is recommended in which 80% of patients reached the parasitological cure rate in the hepatosplenic form that caused by S.mansoni[24,39-41].
Also, it helped in regression of periportal fibrosis. Praziquantel therapy considered ineffective once the late fibrotic stage in the liver and oesophageal variceal bleeding were developed need to be treated by endoscopic therapy or selective shunt procedures[42,43].
Oesophagogastric varices backgrounds
The incidence of first variceal bleeding is 4% per year, with increased risk up to 15% per year in patients with medium and large varices[44]. In 1989, the schistosomal prevalence in Sudan was 6% and increased up to 55% in 2005 at Gezira and Managil which considered as endemic areas[45]. In 2008, oesophageal variceal bleeding was account about 77% of all causes of acute upper gastrointestinal bleeding in a specialized referral center at the capital Khartoum[46]. Schistosomal periportal fibrosis characterized by excess collagen deposition, mainly in the portal tract and disse’s space with obstruction of sinusoidal fenestration, resulting in fibrosis and capillarization of sinusoids which lead progressive occlusion of the portal vein and portal hypertension due to increased intrahepatic vascular resistance and blood flow through the portal venous system[17,47]. Portosystemic collateral veins developed to decompress the portal venous system that manifested clinically by esophageal varices when increasing of the hepatic venous pressure gradient more than 10 mmHg[48-50]. Furthermore, the risk of variceal bleeding increases by other factors like physical exercise, increased intra-abdominal pressure and nonsteroidal anti-inflammatory drugs which alter variceal wall[51-53].
A number of studies for noninvasive oesophageal varices prediction revealed splenomegaly is the most clinical sign[54,55]. As ultrasonography is the preferred initial test which can diagnose the presence of portocollateral vessels[30]. The splenic longitudinal dimension of more than 11 cm and periportal fibrosis of ≥ grade III are significant risk factors for variceal bleeding after at least one bleeding episode in schistosomal portal hypertension patients disease [Figure 5][56]. Computed tomography scan and magnetic resonance imaging (MRI) diagnosed oesophageal varices by a good visualization of the portal venous system. By Angio-MRI the risk of variceal bleeding in patients can be evaluated through azygos blood flow measurement. It’s unlikely to be used in the oesophagogastric variceal screening due to high cost and technical complexity of both imaging tests[57,58].
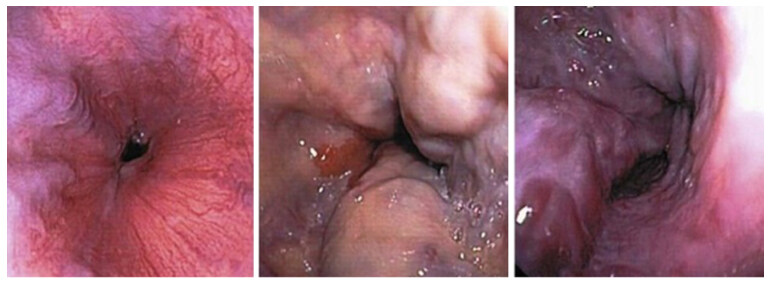
Newly diagnosed patients must undergo screening OGD which considered as a gold standard test for detection of changes that indicate a high risk of bleeding include cherry-red spots, varices on varices, large varices and erosion on varices [Figure 6][59]. Bendtsen et al.[60] classified esophagogastric varices according to size into small varices defined as minimal elevation of veins above the mucosa; while medium varices were known as tortuous variceal veins which occupied less than one-third of the lumen, and large varices occupy more than one-third of the lumen.
Primary prevention of variceal bleeding first episode needs the follow-up of variceal size by the following point[61,62]:
1. If no esophageal varices discovered, screening endoscopy must undergo every three years.
2. If small esophageal varices detected screening endoscopy must be repeated every two years.
3. Medium and large esophageal varices or gastric varices detection need twice daily administration of 80-160 mg propranolol or 80 mg nadolol taken orally with adjustment of the dose if the heart rate of less than 55 beats/min and endoscopic follow-up is not important. Poorly compliance patients or contraindications to beta-blocker drugs need endoscopic band ligation to be considered.
Propanolol reduced portal pressure and blood flow within it through the decreased cardiac output (beta one receptor blockage) and arteriolar splanchnic vasoconstriction (beta two receptor blockage)[63]. Furthermore, it prevents rising of portal pressure during physical exercise and postprandial period and its reducing bacterial translocation rate[64-66]. Some authors have revealed that praziquantel decrease or reverse sonographically periportal fibrosis by parasitic load reduction aimed to primary prophylaxis against a first variceal bleeding in the significant number of schistosomal cases[67-69].
The emergency management of acute variceal bleeding
The mortality rate of the first episode of ruptured variceal bleeding can reach 10%-20% in hepatosplenic schistosomal patients[70]. Besides the schistosomal infection treatment, the severe portal hypertension and it’s complications must be managed similarly to non-parasitic cirrhosis patients including beta-blockers, variceal sclerotherapy or ligation and shunting surgical procedure[71-73].
The association of variceal bleeding with recurrence and death considered as a medical emergency need to be treated in the intensive care unit for hypovolemia correction and complications prevention starting by plasma expander with preference to crystalloid trough at least two 16-gauge peripheral catheters with the regular check of vital signs. Blood samples for baseline laboratory tests can be done upon initial presentation (complete blood count; renal function test; liver function test; international normalized ratio). Furthermore, an introduction of nasogastric tube and urinary catheter for assessment of renal perfusion pressure maintenance and hemodynamic stability indicated by keeping systolic arterial pressure around 90-100 mmHg and cardiac frequency less than < 100 beats/min. Blood transfusion through concentrated erythrocytes to raise hemoglobin level up to 7-9 g/L (hematocrit in 21%-27%) with avoidance of over transfusion that increase portal pressure and risk of recurrent bleeding. While coagulopathy can be corrected by fresh frozen plasma and vitamin K[61,62,74-77].
Airway must be secured immediately especially in encephalopathic patients, due to increased risk of bronchial aspiration of blood or gastric contents during hematemesis and endoscopic procedures. If the airway can’t be maintained then endotracheal intubation is mandatory[78]. Twenty-five percent to fifty percent of patients with variceal bleeding have infections like spontaneous bacterial peritonitis, urinary tract infections and pneumonia. Hence, oral norfloxacin 400 mg twice daily for 7 days was recommended to improve survival and usage of lactulose is mandatory for encephalopathy prevention[61,62,79,80]. Recently intravenous ceftriaxone showed it’s superiority to oral norfloxacin in high-risk patients that suffered from jaundice, ascites, hypovolemic shock and malnutrition[81].
Before diagnostic endoscopy intravenous vasoactive therapy must start urgently with 2 mg every 4 h for the first 48 h and maintained for 5 days using 1 mg of terlipressin every 4 h to prevent rebleeding or somatostatin 250 mg bolus dose followed by 250 mg infused hourly as the second choice for 5 days. Both drugs reduce the hepatic venous pressure gradient, variceal pressure and azygos blood flow[82-87].
Also, terlipressin improves the renal function of hepatorenal syndrome patients by reducing renal vasoconstriction and should not be used in patients with cardiovascular disease[88-92]. Furthermore, another study was showed the use of terlipressin improves outcomes following acute variceal bleeding[93].
Within 12 h of hospital admission OGD performed to identify the source of bleeding and allowing hemostatic treatment done on an empty stomach through nasogastric tube lavage or i.v. erythromycin 250 mg injected 30-60 min before endoscopy[61,62]. Either endoscopic sclerotherapy or endoscopic band ligation can be practiced and the recommendation for endoscopic band ligation as first-line treatment achieved by 5-8 elastic bands maximally per session every 2-3 weeks until the variceal obliteration or small varices ligation become impossible. Complications are fewer than in endoscopic sclerotherapy such as moderate post-ligation ulcer bleeding[61,94]. Several sclerosant agents like ethanol, ethanolamine, polidocanol, sodium morrhuate and tetradecyl sulfate have the similar effect which can be injected paravariceal or intravariceal with 10-30 mL per session every 1-3 weeks until variceal obliteration has been achieved. The endoscopic surveillance required every 3-6 months due to variceal recurrence was seen in 50%-70% of cases[94-96]. The frequent complications include dysphagia, retrosternal pain, bleeding ulcers, esophageal perforation and stricture[94]. The gastric varices rise to 20% in portal hypertensive patients and account for 1%-3% of variceal bleeding which treated by injection of one milliliter of tissue adhesive N-butyl-2-cyanoacrylate three times per session at maximum. With this procedure lung, spleen or brain embolization is the most serious complication[62,97]. Failing of vasoactive drug and endoscopic treatment combination, need initially balloon tamponade followed by repeat endoscopic therapy. If bleeding persists TIPS or surgical shunt creation must be performed as a rescue therapy and second-line treatment[61,94].
A creation of a TIPS percutaneously reduces portal pressure and thus prevents variceal bleeding. It complicated by thrombosis, stenosis and encephalopathy through portal flow diversion from the liver. Selective portosystemic surgical shunt required when TIPS is technically impossible, refractory bleeding and Child-Pugh class A or B patients[98]. Furthermore, liver transplantation considered in Child-Pugh class C patients[61,62,99].
Surgical treatments of refractory variceal bleeding
According to the Baveno V consensus, refractory variceal bleeding was defined as a single episode of clinically significant rebleeding from portal hypertensive sources (recurrent melena, hematemesis resulting in hospital admission, multiple blood transfusion and drop in hemoglobin up to three grams below normal level). Rebleeding within first 5 days was regarded as treatment failure, while rebleeding up to 6 weeks considered as secondary prophylaxis failure[100]. About 15% of asymptomatic patients with varices were expected to bleed over a period of 6 years[101]. Furthermore, patients who survive after the first episode of variceal bleeding have shown a high risk of recurrent bleeding by 60% within 1 year if didn’t have curative therapy while 20% of these patients will die within 4 years[100,102]. There are several factors that associated with variceal rebleeding like red color signs on varices, variceal size, liver disease deterioration, portal pressure, bacterial infection and portal vein thrombosis[103-106]. The rebleeding time frame calcified into very early rebleeding that occur (within 5 days of acute bleeding), early rebleeding (within 6 weeks of acute bleeding) and delayed rebleeding. Therefore, the prevention of variceal rebleeding can start on the sixth day of acute bleeding[107]. The leading cause of death is recurrent variceal bleeding associated with a preserved liver function. Although the end-stage liver failure was rarely seen in schistosomal patients, the deterioration of hepatic reserve has been expected following massive variceal bleeding with developed ascites and the splenic size regression[108,109]. The prevention of recurrent variceal bleeding provided by secondary prophylaxis consists of beta-blocker therapy administration which significantly reduces mortality from 27% to 20% and rebleeding rate from 63% to 42% in treated patients[63,110]. Also, propranolol significantly reduced variceal rebleeding, improved survival and reduces variceal pressure and wall tension in schistosomal portal hypertension[111,112]. Endoscopic band ligation is preferable to endoscopic sclerotherapy by reducing recurrent variceal bleeding risk and variceal stricture incidence in poor intolerance or beta-blocker therapy contraindications[113-117]. Furthermore, other authors have revealed that the combination of beta-blockers and endoscopic band ligation is superior to banding ligation alone in prevention of variceal recurrence and refractory bleeding[118-120]. If both beta-blocker drugs and endoscopic band ligation failed to prevent recurrent variceal bleeding then the options are distal splenorenal shunt or TIPS considered as bridge procedures for liver transplantation[61,62]. The surgical treatment has been considered the best alternative for patients with schistosomal recurrent variceal bleeding due to well-preserved liver function and upper digestive tract bleeding is the dangerous disease complication[121-125]. Currently, Van Buuren et al.[126] showed that sclerotherapy was not effective for variceal bleeding primary prevention. Also, endoscopic therapy has poorer long-term results although it’s less invasive than surgery[126,127]. Furthermore, in 2007 Mudawi and Ibrahim[128] found high rates of variceal rebleeding in 32% of patients with schistosomiasis treated by endoscopic sclerotherapy for secondary prophylaxis and mortality rate was three and a half percent.
Distal splenorenal shunt (Warren’s shunt)
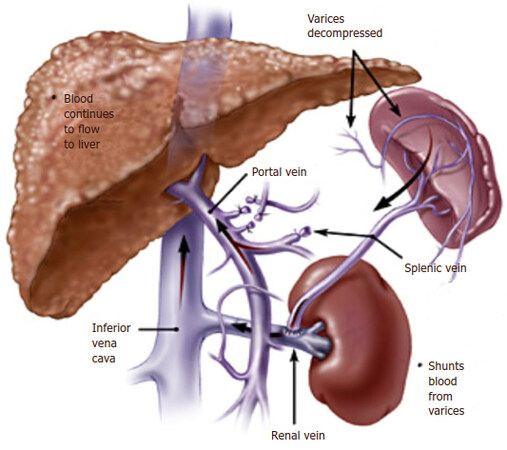
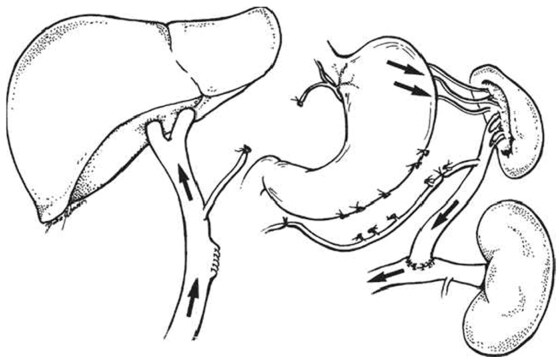
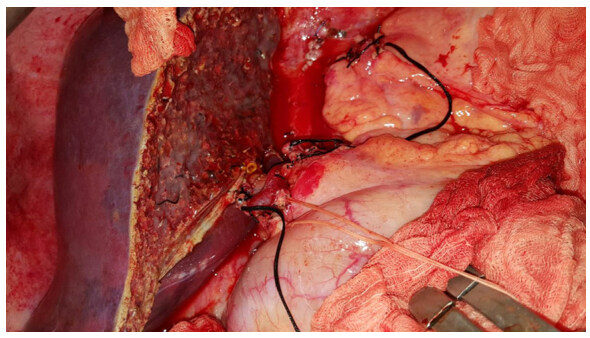
At the University of Miami In 1967, Warren and colleagues[129] were established distal splenorenal shunt (DSRS) aimed to decompress selectively the venous collaterals around the stomach and lower esophagus thereby prevents recurrent variceal bleeding and maintains portal blood flow which reduced the risk of accelerated liver failure and postoperative encephalopathy [Figure 7][129-132]. Most published series was proved the therapeutic efficacy of DSRS with low recurrent variceal bleeding incidence (5%-8%), acceptable risk of hepatic encephalopathy (5%-15%) and excellent long-term survivals[133-135]. The improved surgical techniques in splenopancreatic disconnection and omental division significantly reduced postoperative encephalopathy risk and give good results in patients with schistosomal portal hypertension[123,136-138].
Raia et al.[125] have been demonstrated best results achieved with distal splenorenal shunting for the rate of schistosomal variceal rebleeding in 2.8%-7% of patients. Furthermore, the DSRS improved 5-year survival rate from 80% to 90% particularly in the setting of presinusoidal portal hypertension with adequate liver reserve (Child class A and B)[139-142].
This surgical procedure remains a good option for:
1. Refractory variceal bleeding to pharmacologic and endoscopic therapy with well-preserved liver function unlikely to undergo transplantation within five years[142,143].
2. Portal hypertensive patients with well-preserved liver function and patent splenic vein complicated by refractory bleeding, because of extra-hepatic portal vein thrombosis[142,144].
3. Patients live in some geographic locations who cannot return back for multiple visits of endoscopy or TIPS[142].
4. Also, it’s an effective and reliable technique for selected pediatric cases with portal hypertension not had previous splenectomy and esophageal varices of idiopathic portal hypertension[142,145,146].
Patients who considered for the DSRS needs perioperative evaluation including the following points[142,147]:
1. Upper gastrointestinal endoscopy to confirm high-risk gastroesophageal varices as a source of refractory bleeding.
2. Identify the cause of portal hypertension with variceal bleeding in which hepatosplenic schistosomiasis is the major cause in African countries like Sudan.
3. Imaging of hepatic vascular anatomy required before surgical intervention in which using of doppler ultrasound, CT or magnetic resonance (MR) vascular imaging help in the portal or splenic venous thrombosis identification. In addition, a combination of venous and arterial angiographic study gives information on superior mesenteric, portal and splenic veins patency, flow direction, hepatic venous pressure gradient and left renal vein anatomy .The circumaortic left renal vein was found in 16% of the population, while a total retroaortic left renal vein has been seen in 4% which may preclude performance of DSRS.
4. Evaluation of liver function status by combining the clinical and laboratory parameters to discern the Child-Turcotte-Pugh (CTP) score. Therefore, patients without ascites and CTP score are 7 or 8 (class A or B), considered to have a stable liver disease and candidate for the DSRS procedure.
5. Pulmonary hypertension can occur in 20% of hepatosplenic schistosomiasis (HSS) patients which need preoperative measuring of pulmonary artery pressure and if more than 25 mmHg then DSRS is contraindicated.
Surgical technique of Warren’s shunt
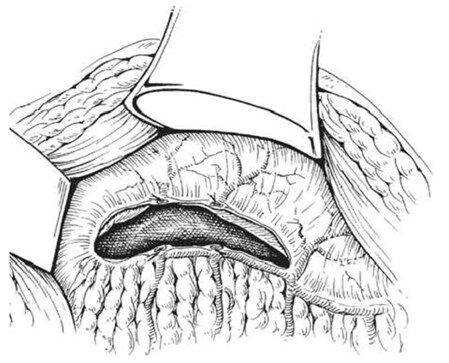
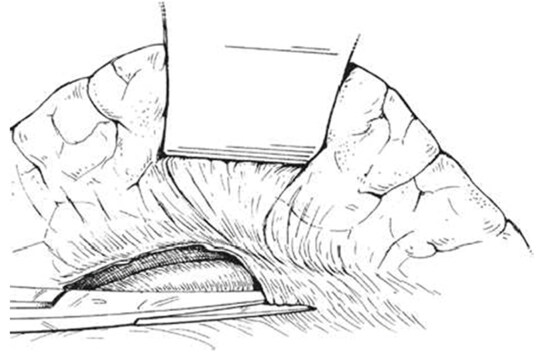
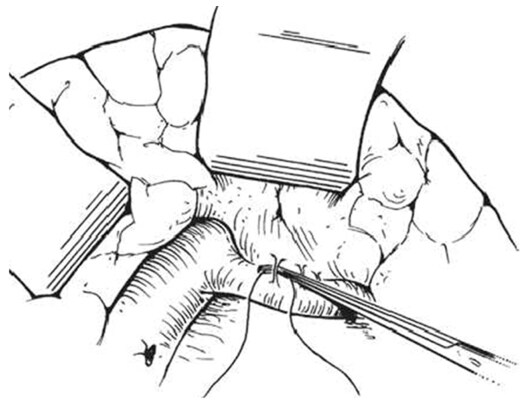
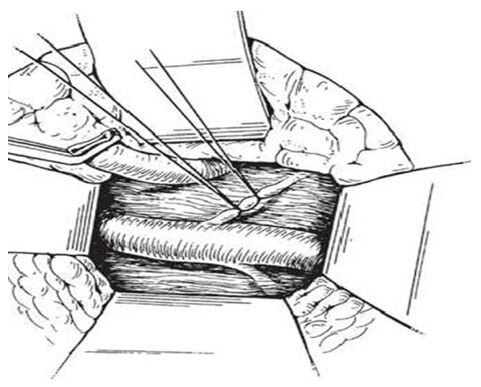
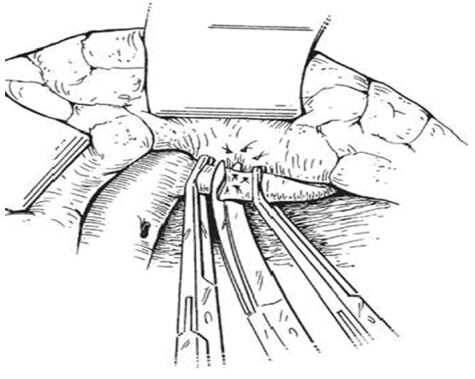
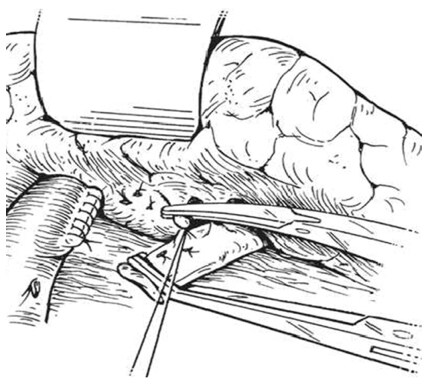
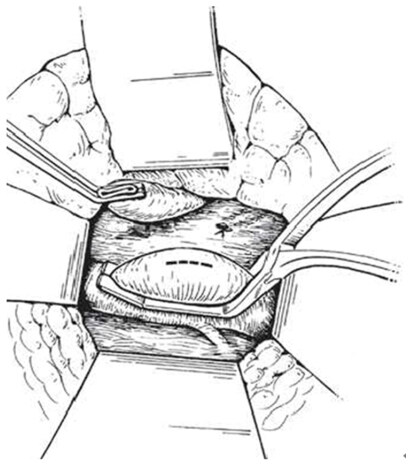
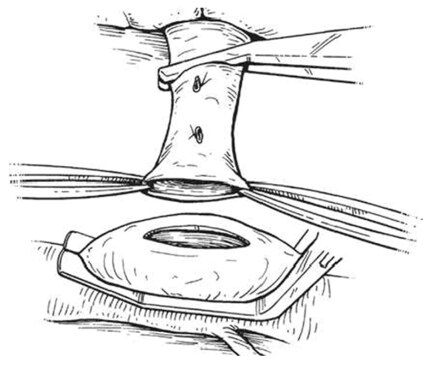
An intraoperative monitoring requires an arterial line, central venous catheter and urinary catheter with the availability of packed red blood cells, fresh-frozen plasma, and platelets in case of major intraoperative blood loss. The patient was lied supine in the operating table with elevated left side slightly and hyperextend it to open an angle between the left lower ribs and iliac crest for exposure of pancreatic tail. The midline abdominal incision extended from xiphisternum to below the umbilicus. Also, the long left subcostal incision is preferable. The peritoneal cavity was opened using coagulating diathermy to achieve hemostasis in dividing tissues and if ascites present must be aspirated and cultured[142]. Well-developed venous collaterals in the falciform ligament must be securely controlled with an assessment of the liver morphology and gallbladder size[148]. The small bowel was isolated in sponge outside the abdominal cavity and the transverse colon retracted inferiorly while the stomach retracted superiorly. Then, through an interruption of the gastroepiploic arcade from the pylorus to the first short gastric vein and the lesser sac was opened to expose the pancreas [Figure 8]. The division of splenocolic ligament and peritoneal incision at the level of the inferior mesenteric vein which traced up towards the pancreas as a guide to identify the splenic vein embedded in the inferior pancreatic border. At the level of the pancreatic neck, an injection of normal saline into soft tissues around splenic vein lifting tissues away from the splenic vein and facilitate its dissection without fear of venous wall damage. Then the splenic vein is dissected initially along its inferior and posterior edge cleared from peritoneum in pancreatic bed and continued medially toward its confluence with superior mesenteric vein until sufficient length was available for coming down to anastomose with left renal vein without kinking [Figure 9]. To avoid the bleeding of the tributaries that arise from the pancreas and entering into the splenic vein, which caused by excessive pancreatic retraction, a fine right-angle clamp is passed around them with the use of 5-0 prolene tie on the vein side and a clip on the pancreatic side [Figure 10][142,148]. The left renal vein was exposed by mobilizing the duodenum after cutting the ligament of Treitz and blunt dissection behind 3rd duodenal part. It desirable to divide left adrenal vein which achieves mobilization of the left renal vein and bring it up into a side-biting vascular clamp for anastomosis without tension with leaving the left gonadal vessel intact which serve as an outflow tract [Figure 11][142,149,150].
Figure 9. The division of posterior peritoneum of the pancreas along its lower border for splenic vein exposure
Figure 10. Ligation of small venous tributaries that arise from the pancreas and entering into the splenic vein
Figure 11. Exposure of the left renal vein by mobilizing the duodenum and left adrenal vein division
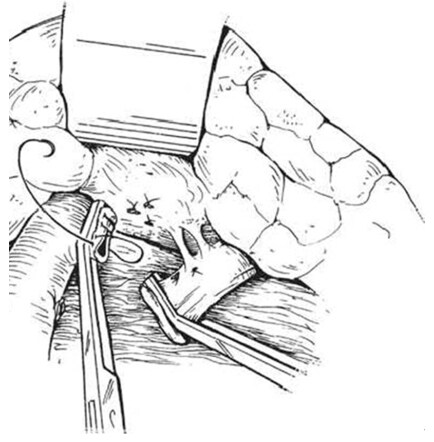
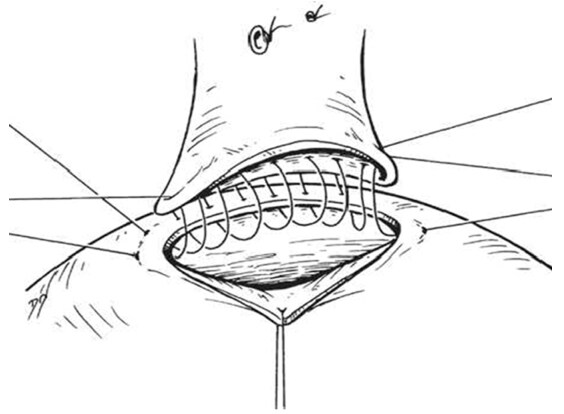
The splenic vein was divided between two vascular clamps after a 2-0 silk ligature was placed near the confluence with superior mesenteric vein and large clip behind the ligature to reduce thrombus formation within the portal vein with oversewing the portal end stump of the splenic vein using 5-0 running prolene [Figures 12 and 13]. Then, the splenic vein trimmed and moves down and Satinsky clamp is applied to the left renal vein opened over sufficient length medially without removing clamps in which both clamps should be held for a tension-free anastomosis [Figures 14 and 15]. After the two veins properly approximated, the stay sutures were placed at either end and anastomosis is made with a running suture of 5-0 prolene in a posterior row while an anterior row of the anastomosis completed with interrupted sutures to prevent a purse-string effect of the running suture [Figures 16 and 17] [144,150]. Once the anastomosis was finished, the completion of porto azygous disconnection by ligation of the umbilical vein and right gastric and coronary veins at lesser curvature while right gastroepiploic vein ligated in continuity near the pyloroduodenal junction. The coronary veins division needs a special care for an avoidance of postoperative hemorrhage or encephalopathy [Figure 18]. Furthermore, to assure long-term shunt patency, the splenic vein diameter is at least 8 mm[149,150].
Figure 14. The sufficient length of splenic vein disconnected from pancreas and moves down to the left renal vein
Figure 17. The stay sutures was placed at either end. Anastomosis is made with sutures of 5-0 prolene in a posterior row and anterior row
The decongestive effect of splenic arterial temporary clamping aid to reduce the large splenic size and congestion in the operative field particularly in schistosomal patients[148]. The abdominal wall closed in two layers by a running non-absorbable suture and according to the literature, no drains were placed due to it enhance secondary bacterial invasion and profound electrolyte disturbances from massive ascetic fluid loss during postoperative drainage of the abdominal cavity[142,148].
Transient postoperative ascites due to retroperitoneal lymphatic vessels manipulation during the dissection of splenic and renal veins which can be reduced by careful fluid and sodium intake with diuretic management[151]. Postoperative infection is another risk factor which decreased by giving the first dose of cephalosporin one hour before DSRS surgery and continued for 48 h after the operation. Ranitidine was given to reduce gastric acidity and albumin 5% is advised within the first 48 h[142,148].
The DSRS patients usually discharged from the hospital 6-8 days after DSRD surgery and maintained in long-term follow up every 6 to 12 months with haemogram, liver function tests to evaluate slightly rise in bilirubin which rarely is greater than 5 mg/dL and doppler ultrasound for shunt patency assessment[142].
Outcomes of Warren’s shunt
A number of recent clinical studies were compared surgical shunting with TIPS in the management of refractory variceal bleeding reported that DSRS has significantly lower rebleeding rate and encephalopathy than TIPS[152]. In Child-Pugh class A and B patients Henderson et al.[153] was compared distal splenorenal shunt with TIPS and revealed that rebleeding risk was higher in TIPS (5.5% vs. 9%) and the re-intervention rate was significantly higher in the TIPS (11% vs. 82%) respectively.
Other studies were showed that the incidence of recurrent variceal bleeding post-DSRS is 3.8%-14% while 15%-30% following TIPS[154-161]. The postoperative DSRS encephalopathy rate is 5%-19%[154-158,162].
The lower incidence of post-selective shunt encephalopathy in the schistosomal patients as compared to that in cirrhotics, 12% in the first 5 years and up to 27% at 10 years[163,164]. Spina et al.[141] has been compared the Warren shunt with endoscopic sclerotherapy and showed the Warren shunt improve survival and reduced rebleeding risk without worsening encephalopathy.
Another study was revealed the recurrent bleeding after splenectomy with devascularization was 27%[165]. According to the literature, the mortality rates following DSRS is 2.2%-14.8% in patients with schistosomal portal hypertension[166,125].
Liver transplantation
In 1967, the first successful hepatic transplantation was reported by Starzl et al.[167] in the United State. Then one year later in Cambridge Calne et al.[168] performed the first human orthotopic liver transplantation in Europe. Since that time its rapidly becoming the standard therapy for end-stage liver diseases with significantly improved survival rates post-transplant achieving 96% at the first year and 71% at the tenth year[169].
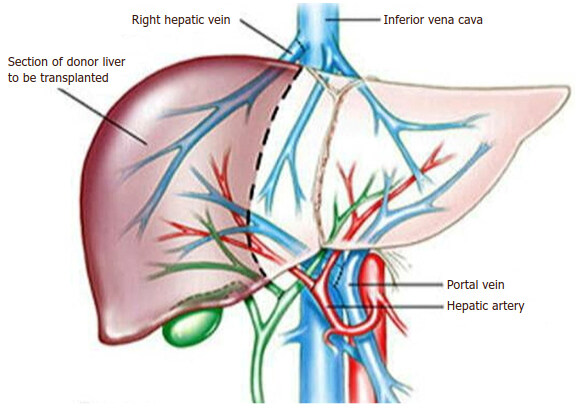
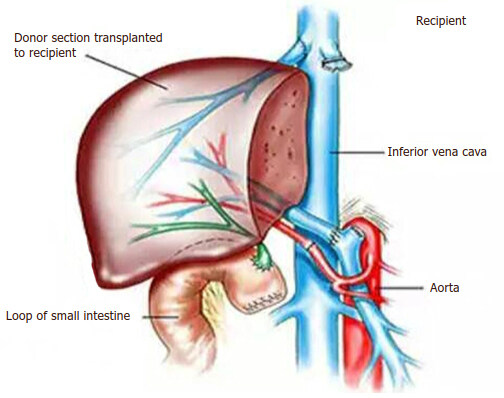
Recently liver transplantation becomes successful because of improvement in surgical techniques, organ preservation, immunosuppression and critical care which means potential recipients is rising in number, with donor graft reduction in many countries. A trial to increase the donor pool by split livers, selected brain-stem death donors, and living donors[170]. The living donors were considered as a source of liver grafts potentially from brothers, sisters or spouses in Asian countries such as Japan which associated with the severely restricted availability of deceased donors [Figures 19 and 20] [171].
The United Network for Organ Sharing in the USA was showed the usage of living-donor liver grafts reduced from ten percent in 2001 to 3.9% in 2008 while a rise in deceased donors from 0.5% in 1998 to 5% in 2007[172].
On the other hand, the hepatic living donor grafts have three distinct advantages when compared with deceased donor grafts including of better graft function, shorter cold-ischemia time and help in early transplantation [Figures 21 and 22] [173]. The vast majority of patients with significant portal hypertension who require liver transplants aimed specifically to treat one of its major complications, namely, oesophageal variceal bleeding. Therefore, it has a great potential to control recurrent bleeding episodes and correction of portal hypertension[174-176]. Some authors were revealed that the liver transplantation in schistosomal portal hypertension with preserved hepatic function has been indicated in recurrent variceal bleeding, patients who may develop advanced liver dysfunction predicted by (Child-Pugh score > 8 or model for end-stage liver disease score, MELD > 16) and failure of the DSRS procedure because of excessive shunt tension, kinking or twisting and renal venous hypertension secondary to left renal vein tenting[99,177-179]. Vincenzi et al.[180] and Pannegeon et al.[181] reported some cases of infected liver grafts by schistosomiasis from living or deceased donors and the recipients was treated by anti-schistosomal drugs and did well after hepatic transplantation with functioning grafts without infection reactivation risk and this associated with a favorable long-term outcome. Sometimes, a large number of cases with variceal bleeding cannot be a candidate for liver transplantation as an emergency procedure because of advanced age, non-compliance for immunosuppressive drugs, advanced comorbidities and long waiting list time related to donor graft shortage[176,179,182]. Hence, with the presence of hepatic reserve adequacy and hemodynamic stability, the emergent selective shunt can be used with a high success rate[175,179]. In developing countries like Sudan, the liver transplantation was not established yet because it’s always interfaced with needs to resolve a number of medical and organizational issues, comes from the government (juristic and legislative-based), as well as from the practical healthcare (hospital equipments, human resources)[183]. Furthermore, the cultural difficulties of organ donation post-brain death require more discussion by individuals and society for more response[183].
In conclusion, this review has demonstrated that Warren’s shunt is an excellent alternative approach to the liver transplantation in the treatment of schistosomal refractory variceal bleeding especially for the developing countries where the facilities for liver transplantation and postoperative care are not available or good enough. It has very good results with low operative morbidity and mortality. Therefore, it improved the survival rate and provides a long-term management.
Declarartions
AcknowledgmentsThe authors would like to thank Prof. Wei-Chen Lee for his unlimited support and kind suggestions.
Authors’ contributionsContributed to writing, design, critical revision of contents, drafting and final approval of the manuscript: Doush WMA
Contributed to revision and final approval of the manuscript to be published: Elzein JA
Read and approved the final manuscript: Doush WMA, Elzein JA
Financial support and sponsorshipNone.
Conflicts of interestThere are no conflicts of interest.
Patient consentNot applicable.
Ethics approvalNot applicable.
Copyright© The Author(s) 2018.
REFERENCES
1. D'Amico G, De Franchis R; Cooperative Study Group. Upper digestive bleeding in cirrhosis. Post-therapeutic outcome and prognostic indicators. Hepatology 2003;38:599-612.
2. De Franchis R, Primignani M. Natural history of portal hypertension in patients with cirrhosis. Clin Liver Dis 2001;5:645-63.
3. Lai YS, Biedermann P, Ekpo UF, Garba A, Mathieu E, Midzi N, Mwinzi P, N'Goran EK, Raso G, Assaré RK, Sacko M. Spatial distribution of schistosomiasis and treatment needs in sub-Saharan Africa: a systematic review and geostatistical analysis. Lancet Infects Dis 2015;15:927-40.
4. Khamis H, Abdul Wahab SM. An overview of Mohammed Salih Idris (MSI) center for acute gastrointestinal bleeding: a look into one year experience in acute GI bleeding. Sudan Medical Monitor 2008;3:135-38.
5. Kheir MM, Eltoum IA, Saad AM, Ali MM, Baraka OZ, Homeida MM. Mortality due to schistosomiasis mansoni: a field study in Sudan. Am J Trop Med Hyg 1999;60:307-10.
6. Ferraz AA, Bacelar T, Silveira MJ, Coelho AR, Câmara NR, de Araújo JJ, Ferraz EM. Surgical treatment of schistosomal portal hypertension. Int Surg 2000;86:1-8.
7. Lo GH, Lai KH, Ng WW, Tam TN, Lee SD, Tsai YT, Lo KJ. Injection sclerotherapy preceded by esophageal tamponade versus immediate sclerotherapy in arresting active variceal bleeding: a prospective randomized trial. Gastrointest Endosc 1992;38:421-4.
8. Garcia-Tsao G, Sanyal AJ, Grace ND, Carey W. Prevention and management of gastroesophageal varices and variceal hemorrhage in cirrhosis. Hepatology 2007;46:922-38.
9. D'amico G, Pagliaro L, Bosch J. The treatment of portal hypertension: a meta-analytic review. Hepatology 1995;22:332-54.
10. Badr M. Detection of bladder cancer. In: El-Bolkany C, editor. Bladder cancer Al-Ahram Press; 1981. p. 1.
14. Christopherson JB. Intravenous injections of antimonium tartaratum in bilharziosis. Br Med J 1918;2:652-3.
15. Rollemberg CV, Santos CM, Silva MM, Souza AM, Silva AM, Almeida JA, Almeida RP. Epidemiological characteristics and geographical distribution of schistosomiasis and geohelminths, in the State of Sergipe, according to data from the Schistosomiasis Control Program in Sergipe. Rev Soc Bras Med Trop 2011;44:91-6.
16. World Health Organization. The control of schistosomiasis: second report of the WHO expert committee. WHO Tech Rep Seri 1993;830:1-86.
17. Grimaud JA, Borojevic R. Chronic human schistosomiasis mansoni. Pathology of the Disse's space. Lab Invest 1977;36:268-73.
18. Gray DJ, Ross AG, Li YS, McManus DP. Diagnosis and management of schistosomiasis. BMJ 2011;342:d2651.
19. Chistulo L, Loverde P, Engels D. Disease watch: Schistosomiasis. Nat Rev Microbiol 2004;2:12.
20. Ross AG, Bartley PB, Sleigh AC, Olds GR, Li Y, Williams GM, McManus DP. Schistosomiasis. N Engl J Med 2002;346:1212-20.
21. Warren KS. The pathology, pathobiology, and pathogenesis of schistosomiasis. Nature 1978;273:609-12.
22. Madwar MA, El Tahawy M, Strickland GT. The relationship between uncomplicated schistosomiasis and hepatitis B infection. Trans R Soc Trop Med Hyg 1989;83:233-236.
23. Richter J, Correia Dacal AR, Vergetti Siqueira JG, Poggensee G, Mannsmann U, Deelder A, Feldmeier H. Sonographic prediction of variceal bleeding in patients with liver fibrosis due to Schistosoma mansoni. Trop Med Int Health 1998;3:728-35.
24. Vitorino RR, Souza FPC, Costa AP, Júnior FCF, Santana LA, Gomes AP. Schistosomiasis mansoni: diagnosis, treatment, epidemiology, prophylaxis, control. Rev Bras Clin Med 2012;10:39-45.
25. Nash TE, Cheever AW, Ottesen EA, Cook JA. Schistosome infections in humans: perspectives and recent findings. Ann Intern Med 1982;97:740-54.
26. Leite LA, Pimenta Filho AA, da Fonseca CS, dos Santos BS, dos Santos Ferreira RD, Montenegro SM, Lopes EP, Domingues AL, Owen JS, de Menezes Lima VL. Hemostatic dysfunction is increased in patients with hepatosplenic schistosomiasis mansoni and advanced periportal fibrosis. PLoS Negl Trop Dis 2013;7:e2314.
27. Alves A Jr, Fontes DA, Melo VA, Machado MC, Cruz JF, Santos EA. Schistosomal portal hypertension: influence of the portal blood flow in serum levels of hepatic enzymes. Arq Gastroenterol 2003;40:203-8.
28. Camacho-Lobato L, Borges DR. Early liver dysfunction in schistosomiasis. J Hepatol 1998;29:233-40.
29. Omran SA, Amin HM, El-Bassiouni NE, Essawy FM, Toiema SM. Vitamin K dependent coagulation proteins in endemic hepatosplenomegaly in Egypt. J Clin Pathol 1994;47:502-4.
30. Zoli M, Iervese T, Merkel C, Bianchi G, Magalotti D, Marchesini G, Gatta A, Pisi E. Prognostic significance of portal hemodynamics in patients with compensated cirrhosis. J Hepatol 1993;17:56-61.
31. Homeida M, Abdel-Gadir AF, Cheever AW, Bennett JL, Arbab BM, Ibrahium SZ, Abdel-Salam IM, Dafalla AA, Nash TE. Diagnosis of pathologically confirmed Symmers' periportal fibrosis by ultrasonography: a prospective blinded study. Am J Trop Med Hyg 1988;38:86-91.
32. Abdel-Wahab MF, Esmat G, Farrag A, El-Boraey YA, Strickland GT. Grading of hepatic schistosomiasis by the use of ultrasonography. Am J Trop Med Hyg 1992;46:403-8.
33. Paranagua-Vezozzo DC, Cerri GG. Duplex hemodynamic evaluation of hepatosplenic mansoni schistosomiasis. Mem Inst Oswaldo Cruz 1992;87:149-51.
34. Schwartz BS, Mawhorter SD. Parasitic infections in solid organ transplantation. Am J Transplant 2013;13:280-303.
35. Strauss E. Hepatosplenic schistosomiasis: a model for the study of portal hypertension. Ann Hepatol 2001;1:6-11.
36. Ahmed K, Safdar K, Kemmer N, Atiq M, Wang J, Neff GW. Intestinal schistosomiasis following orthotopic liver transplantation: a case report. Transplant Proc 2007;39:3502-4.
37. Hoare M, Gelson WT, Davies SE, Curran M, Alexander GJ. Hepatic and intestinal schistosomiasis after orthotopic liver transplant. Liver Transpl 2005;11:1603-7.
38. Kamal SM, El Sayed Khalifa K. Immune modulation by helminthic infections: worms and viral infections. Parasite Immunol 2006;28:483-96.
39. Mahmoud AA. Praziquantel for the treatment of helminthic infections. Ad Intern Med 1986;32:193-206.
40. Domingues AL, Coutinho AD. Reduction of morbidity in hepatosplenic schistosomiasis mansoni after treatment with praziquantel: a long term study. Rev Soc Bras Med Trop 1990;23:101-7.
41. Montresor A, Engels D, Chitsulo L, Bundy DA, Brooker S, Savioli L. Development and validation of a 'tablet pole' for the administration of praziquantel in sub-Saharan Africa. Trans R Soc Trop Med Hyg 2001;95:542-4.
42. Elliott DE. Schistosomiasis: pathophysiology, diagnosis, and treatment. Gastroenterol Clin North Am 1996;25:599-625.
43. Soentjens P, Clerinx J. Diagnosis of schistosomiasis. Available from: https://www.uptodate.com/contents/diagnosis-of-schistosomiasis?search=Diagnosis%20and%20treatment%20of%20schistosomiasis&source=search_result&selectedTitle=3~78&usage_type=default&display_rank=3. [Last accessed on 16 Jan 2018].
44. D'Amico G, Luca A. Natural history-clinical-haemodynamic correlations: prediction of the risk of bleeding. Baillieres Clin Gastroenterol 1997;11:243-56.
45. Alla GN. Results of the 2005 surveillance project in Gezira. Federal Ministry of Health Report 2009.
46. Khamis H. An overview of Mohammed Salih Idris (MSI) center for acute upper gastrointestinal (GI) bleeding: a look into one year experience in acute GI bleeding. Sudan Medical Monitor 2008;3:138-8.
47. Burke ML, Jones MK, Gobert GN, Li YS, Ellis MK, McManus DP. Immunopathogenesis of human schistosomiasis. Parasite Immunol 2009;31:163-76.
48. Bosch J, Mastai R, Kravetz D, Navasa M, Rodes J. Hemodynamic evaluation of the patient with portal hypertension. Semin Liver Dis 1986;6:309-17.
49. Garcia-Tsao G, Groszmann RJ, Fisher RL, Conn HO, Atterbury CE, Glickman M. Portal pressure, presence of gastroesophageal varices and variceal bleeding. Hepatology 1985;5:419-24.
50. Nahum H, Benhamou JP. Portal hypertension, size of esophageal varices, and risk of gastrointestinal bleeding in alcoholic cirrhosis. Gastroenterology 1980;79:1139-44.
51. Garcia-Pagan JC, Santos CR, Barbera JA, Luca A, Roca JO, Rodriguez-Roisin RO, Bosch JA, Rodes JO. Physical exercise increases portal pressure in patients with cirrhosis and portal hypertension. Gastroenterology 1996;111:1300-6.
52. Luca A, Cirera I, García-Pagán JC, Feu F, Pizcueta P, Bosch J, Rodés J. Hemodynamic effects of acute changes in intra-abdominal pressure in patients with cirrhosis. Gastroenterology 1993;104:222-7.
53. De Ledinghen V, Heresbach D, Fourdan O, Bernard P, Liebaert-Bories MP, Nousbaum JB, Gourlaouen A, Becker MC, Ribard D, Ingrand P, Silvain C, Beauchant M. Anti-inflammatory drugs and variceal bleeding: a case-control study. Gut 1999;44:270-3.
54. Giannini E, Botta F, Borro P, Risso D, Romagnoli P, Fasoli A, Mele MR, Testa E, Mansi C, Savarino V, Testa R. Platelet count/spleen diameter ratio: proposal and validation of a non-invasive parameter to predict the presence of oesophageal varices in patients with liver cirrhosis. Gut 2003;52:1200-5.
55. Sanyal AJ, Fontana RJ, Di Bisceglie AM, Everhart JE, Doherty MC, Everson GT, Donovan JA, Malet PF, Mehta S, Sheikh MY, Reid AE. The prevalence and risk factors associated with esophageal varices in subjects with hepatitis C and advanced fibrosis. Gastrointest Endosc 2006;64:855-64.
56. Eltoum IA, Taha TE, Saad AM, Suliman SM, Bennett JL, Nash TE, Homeida MM. Predictors of upper gastrointestinal bleeding in patients with schistosomal periportal fibrosis. Br J Surg 1994;81:996-9.
57. Cho KC, Patel YD, Wachsberg RH, Seeff J. Varices in portal hypertension: evaluation with CT. Radiographics 1995;15:609-22.
58. Johnson CD, Ehman RL, Rakela J, Ilstrup DM. MR angiography in portal hypertension: detection of varices and imaging techniques. J Comput Assist Tomogr 1991;15:578-84.
59. Spence RA. Prevention of recurrent bleeding: nonshunting surgery Portal hypertension. Japan: Springer; 1991. pp. 533-47.
60. Bendtsen F, Skovgaard LT, Sørensen TI, Matzen P. Agreement among multiple observers on endoscopic diagnosis of esophageal varices before bleeding. Hepatology 1990;11:341-7.
61. Lebrec D, Vinel JP, Dupas JL. Complications of portal hypertension in adults: a French consensus. Eur J Gastroenterol Hepatol 2005;17:403-10.
62. De Franchis R. Evolving Consensus in Portal Hypertension. Report of the Baveno IV consensus workshop on methodology of diagnosis and therapy in portal hypertension. J Hepatol 2005;43:167-76.
63. Garcia-Tsao G. Current management of the complications of cirrhosis and portal hypertension: variceal hemorrhage, ascites, and spontaneous bacterial peritonitis. Gastroenterology 2001;120:726-48.
64. Bandi JC, Garcia-Pagan JC, Escorsell A, François E, Moitinho E, Rodés J, Bosch J. Effects of propranolol on the hepatic hemodynamic response to physical exercise in patients with cirrhosis. Hepatology 1998;28:677-82.
65. Perez-Paramo M, Munoz J, Albillos A, Freile I, Portero F, Santos M, Ortiz-Berrocal J. Effect of propranolol on the factors promoting bacterial translocation in cirrhotic rats with ascites. Hepatology 2000;31:43-8.
66. Bendtsen F, Simonsen L, Henriksen JH. Effect on hemodynamics of a liquid meal alone and in combination with propranolol in cirrhosis. Gastroenterology 1992;102:1017-23.
67. Homeida MA, El Tom I, Nash T, Bennett JL. Association of the therapeutic activity of praziquantel with the reversal of Symmers' fibrosis induced by Schistosoma mansoni. Am J Trop Med Hyg 1991;45:360-5.
68. Doehring-Schwerdtfeger E, Abdel-Rahim IM, Kardoff R, Kaiser C, Franke D, Schlake J. Ultrasonographical investigation of periportal fibrosis in children with Schistosoma mansoni infection: reversibility of morbidity twenty-three months after treatment with praziquantel. Am J Trop Med Hyg 1992;46:409-15.
69. Berhe N, Myrvang B, Gundersen SG. Reversibility of schistosomal periportal thickening/fibrosis after praziquantel therapy: a twenty-six month follow-up study in Ethiopia. Am J Trop Med Hyg 2008;78:228-34.
70. Makdissi FF, Herman P, Pugliese V, de Cleva R, Saad WA, Cecconello I, D'Albuquerque LA. Long-term results of esophagogastric devascularization and splenectomy associated with endoscopic treatment in schistosomal portal hypertension. World J Surg 2010;34:2682-8.
71. Da Silva LC, Strauss E, Gayotto LC, Mies S, Macedo AL, da Silva AT, Silva EF, Lacet CM, Antonelli RH, Fermanian J. A randomized trial for the study of the elective surgical treatment of portal hypertension in mansonic schistosomiasis. Ann Surg 1986;204:148-53.
72. Andersson KL, Chung RT. Hepatic schistosomiasis. Curr Treat Options Gastroenterol 2007;10:504-12.
73. Raia S, Da Silva LC, Gayotto LC, Forster SC, Fukushima J, Strauss E. Portal hypertension in schistosomiasis: a long-term follow-up of a randomized trial comparing three types of surgery. Hepatology 1994;20:398-403.
74. Bittencourt PL, Farias AQ, Strauss E, Mattos AA. Variceal bleeding: consensus meeting report from the Brazilian Society of Hepatology. Arq Gastroenterol 2010;47:202-16.
75. Duggan JM. Review article: transfusion in gastrointestinal hemorrhage-If, when and how much? Aliment Pharmacol Ther 2001;15:1109-13.
76. McCormick PA, Jenkins SA, McIntyre N, Burroughs AK. Why portal hypertensive varices bleed and bleed: a hypothesis. Gut 1995;36:100-3.
77. Castaneda B, Debernardi-Venon W, Bandi JC, Andreu V, Pérez-del-Pulgar S, Moitinho E, Pizcueta P, Bosch J. The role of portal pressure in the severity of bleeding in portal hypertensive rats. Hepatology 2000;31:581-6.
78. Cárdenas A, Ginès P, Uriz J, Bessa X, Salmerón JM, Mas A, Ortega R, Calahorra B, De Las Heras D, Bosch J, Arroyo V. Renal failure after upper gastrointestinal bleeding in cirrhosis: incidence, clinical course, predictive factors, and short-term prognosis. Hepatology 2001;34:671-6.
79. Bleichner G, Boulanger R, Squara P, Sollet JP, Parent A. Frequency of infections in cirrhotic patients presenting with acute gastrointestinal haemorrhage. Br J Surg 1986;73:724-6.
80. Rimola A, García-Tsao G, Navasa M, Piddock LJ, Planas R, Bernard B, Inadomi JM. Diagnosis, treatment and prophylaxis of spontaneous bacterial peritonitis: a consensus document. J Hepatol 2000;32:142-53.
81. Fernández J, del Arbol LR, Gómez C, Durandez R, Serradilla R, Guarner C, Planas R, Arroyo V, Navasa M. Norfloxacin vs ceftriaxone in the prophylaxis of infections in patients with advanced cirrhosis and hemorrhage. Gastroenterology 2006;131:1049-56.
82. Nevens F, Van Steenbergen W, Yap SH, Fevery J. Assessment of variceal pressure by continuous non-invasive endoscopic registration: a placebo controlled evaluation of the effect of terlipressin and octreotide. Gut 1996;38:129-34.
83. Moreau R, Soubrane O, Hadengue A, Sogni P, Gaudin C, Kleber G, Lebrec D. Hemodynamic effects of the administration of terlipressin alone or combined with nitroglycerin in patients with cirrhosis. Gastroenterol Clin Biol 1991;16:680-6.
84. Bosch J, Kravetz D, Rodes J. Effects of somatostatin on hepatic and systemic hemodynamics in patients with cirrhosis of the liver: comparison with vasopressin. Gastroenterology 1981;80:518-25.
85. Cirera I, Feu F, Luca A, García-Pagán JC, Fernández M, Escorsell A, Bosch J, Rodés J. Effects of bolus injections and continuous infusions of somatostatin and placebo in patients with cirrhosis: a double-blind hemodynamic investigation. Hepatology 1995;22:106-11.
86. Villanueva C, Ortiz J, Miñana J, Soriano G, Sàbat M, Boadas J, Balanzó J. Somatostatin treatment and risk stratification by continuous portal pressure monitoring during acute variceal bleeding. Gastroenterology 2001;121:110-7.
87. Nevens F, Sprengers D, Fevery J. The effect of different doses of a bolus injection of somatostatin combined with a slow infusion on transmural oesophageal variceal pressure in patients with cirrhosis. J Hepatol 1994;20:27-31.
88. Bosch J, Dell'era A. Vasoactive drugs for the treatment of bleeding esophageal varices. Gastroenterol Clin Biol 2004;28:B186-9.
89. Moreau R, Durand F, Poynard T, Duhamel C, Cervoni JP, Ichaï P, Abergel A, Halimi C, Pauwels M, Bronowicki JP, Giostra E. Terlipressin in patients with cirrhosis and type 1 hepatorenal syndrome: a retrospective multicenter study. Gastroenterology 2002;122:923-30.
90. Ortega R, Gines P, Uriz J, Cárdenas A, Calahorra B, De Las Heras D, Guevara M, Bataller R, Jiménez W, Arroyo V, Rodés J. Terlipressin therapy with and without albumin for patients with hepatorenal syndrome: results of a prospective, nonrandomized study. Hepatology 2002;36:941-8.
91. Uriz J, Ginès P, Cárdenas A, Sort P, Jiménez W, Salmerón JM, Bataller R, Mas A, Navasa M, Arroyo V, Rodés J. Terlipressin plus albumin infusion: an effective and safe therapy of hepatorenal syndrome. J Hepatol 2000;33:43-8.
92. Feu FA, Del Arbol LR, Banares RA, Planas RA, Bosch JA. Double-blind randomized controlled trial comparing terlipressin and somatostatin for acute variceal hemorrhage. Variceal Bleeding Study Group. Gastroenterology 1996;111:1291-9.
93. D'Amico G, Pietrosi G, Tarantino I, Pagliaro L. Emergency sclerotherapy versus vasoactive drugs for variceal bleeding in cirrhosis: a Cochrane metaanalysis. Gastroenterology 2003;124:1277-91.
94. Luketic VA. Management of portal hypertension after variceal hemorrhage. Clin Liver Dis 2001;5:677-707.
95. Waked I, Korula J. Analysis of long-term endoscopic surveillance during follow-up after variceal sclerotherapy from a 13-year experience. Am J Med 1997;102:192-9.
96. Westaby D, Macdougall BR, Williams R. Improved survival following injection sclerotherapy for esophageal varices: final analysis of a controlled trial. Hepatology 1985;5:827-30.
97. Sarin SK, Lahoti D, Saxena SP, Murthy NS, Makwana UK. Prevalence, classification and natural history of gastric varices: a long-term follow-up study in 568 portal hypertension patients. Hepatology 1992;16:1343-9.
98. Henderson JM. Salvage therapies for refractory variceal hemorrhage. Clin Liver Dis 2001;5:709-25.
99. De Franchis R. Evolving consensus in portal hypertension: report of the Baveno IV consensus workshop on methodology of diagnosis and therapy in portal hypertension. J Hepatol 2005;43:167-76.
100. De Franchis R. Revising consensus in portal hypertension: report of the Baveno V consensus workshop on methodology of diagnosis and therapy in portal hypertension. J Hepatol 2010;53:762-8.
101. Ibrahim SZ, Shah TI, Arbab BM, Abdelwahab O. Risk factors for bleeding in patients with asymptomatic varices secondary to schistosomal portal hypertension. Sudan Med J 2009;45:35-41.
102. Ibrahim SZ, Suliman SH, Arbab BM, Abdelwahab O, Elmasri SH, Hassan MA. The natural history of bleeding oesophageal varices in patients with schistosomal portal hypertension. Khartoum Med J 2010;3:382-92.
103. Mihas AA, Sanyal AJ. Recurrent variceal bleeding despite endoscopic and medical therapy. Gastroenterology 2004;127:621-9.
104. Lo GH, Lai KH. The factors affecting risk of recurrent variceal bleeding. Gastroenterology 2005;128:244-5.
105. Hou MC, Lin HC, Kuo BI, Liao TM, Lee FY, Chang FY, Lee SD. Sequential variceal pressure measurement by endoscopic needle puncture during maintenance sclerotherapy: the correlation between variceal pressure and variceal rebleeding. J Hepatol 1998;29:772-8.
106. Dell'era A, Bosch J. The relevance of portal pressure and other risk factors in acute gastro-oesophageal variceal bleeding. Aliment Pharmacol Ther 2004;20:8-16.
107. Burroughs AK, Calès P, Kravetz D. Definition of key events-last attempt? In portal hypertension IV. Proceedings of the Fourth Baveno International Consensus Workshop on Methodology of Diagnosis and Treatment. Oxford: Blackwell Science; 2006. pp. 11-39.
108. Da Silva LC, Carrilho FJ. Hepatosplenic schistosomiasis. Pathophysiology and treatment. Gastroenterol Clin North Am 1992;21:163-77.
109. Gasim B, Fedail SS, Musaad AM, Salih SM, Ibn-ouf M. Endoscopic sclerotherapy for bleeding oesophageal varices: experience in Sudan. Trop Gastroenterol 2001;23:107-9.
110. D'Amico G, Pagliaro L, Bosch J. Pharmacological treatment of portal hypertension: an evidence-based approach. Semin Liver Dis 1999;19:475-505.
111. Tourabi HE, Amin AE, Shaheen M, Woda SA, Homeida M, Harron DW. Propranolol reduces mortality in patients with portal hypertension secondary to schistosomiasis. Ann Trop Med Parasitol 1994;88:493-500.
112. Farias AQ, Kassab F, da Rocha EC, Dos Santos Bomfim V, Vezozzo DC, Bittencourt PL, Carrilho FJ. Propranolol reduces variceal pressure and wall tension in schistosomiasis presinusoidal portal hypertension. J Gastroenterol Hepatol 2009;24:1852-6.
113. D'Amico G, Pagliaro L, Bosch J. The treatment of portal hypertension: a meta-analytic review. Hepatology 1995;22:332-54.
114. Heresbach D, Jacquelinet C, Nouel O, Chaperon J, Bretagne JF, Gosselin M. Sclerotherapy versus ligation in hemorrhage caused by rupture of esophageal varices. Direct meta-analysis of randomized trials. Gastroenterol Clin Biol 1995;19:914-20.
115. Laine L, Cook D. Endoscopic ligation compared with sclerotherapy for treatment of esophageal variceal bleeding. A meta-analysis. Ann Intern Med 1995;123:280-7.
116. Stiegmann GV, Goff JS, Sun JH, Davis D, Silas D. Technique and early clinical results of endoscopic variceal ligation (EVL). Surg Endosc 1989;3:73-8.
117. Gimson AE, Ramage JK, Panos MZ, Hayllar K, Harrison PM, Williams R, Westaby D. Randomised trial of variceal banding ligation versus injection sclerotherapy for bleeding oesophageal varices. Lancet 1993;342:391-4.
118. Lo GH, Lai KH, Cheng JS, Chen MH, Huang HC, Hsu PI, Lin CK. Endoscopic variceal ligation plus nadolol and sucralfate compared with ligation alone for the prevention of variceal rebleeding: a prospective, randomized trial. Hepatology 2000;32:461-5.
119. De la Pena J, Brullet E, Sanchez-Hernández E, Sanchez-Hernández E, Rivero M, Vergara M, Martin-Lorente JL, Garcia Suárez C. Variceal ligation plus nadolol compared with ligation for prophylaxis of variceal rebleeding: a multicenter trial. Hepatology 2005;41:572-8.
120. Boyer TD. Pharmacologic treatment of portal hypertension: past, present, and future. Hepatology 2001;34:834-9.
121. Ferraz AA, Bacelar T, Silveira MJ, Coelho AR, Camara NR, de Araújo JJ, Ferraz EM. Surgical treatment of schistosomal portal hypertension. Int Surg 2000;86:1-8.
122. Ezzat FA, Abu-Elmagd KM, Aly MA, Fathy OM, El-ghawlby NA, El-fiky AM, el-Barbary MH. Selective shunt versus nonshunt surgery for management of both schistosomal and nonschistosomal variceal bleeders. Ann Surg 1990;212:97-108.
123. Ezzat FA, Abu-Elmagd KM, Aly IY, Aly MA, Fathy OM, el-Barbary MH, Bahgat OO, Salam AA, Kutner MH. Distal splenorenal shunt for management of variceal bleeding in patients with schistosomal hepatic fibrosis. Ann Surg 1986;204:566-73.
124. Sakai P, Boaventura S, Capacci ML, Macedo TM, Ishioka SZ. Endoscopic sclerotherapy of bleeding esophageal varices: a comparative study of results in patients with schistosomiasis and cirrhosis. Endoscopy 1988;20:134-6.
125. Raia S, Da Silva LC, Gayotto LCC, Forster SC, Fukushima J, Strauss E. Portal hypertension in schistosomiasis: a long-term follow-up of a randomized trial comparing three types of surgery. Hepatology 1994;20:398-403.
126. Van Buuren HR, Rasch MC, Batenburg PL, Bolwerk CJ, Nicolai JJ, van der Werf SD, Scherpenisse J, Arends LR, van Hattum J, Rauws EA, Schalm SW. Endoscopic sclerotherapy compared with no specific treatment for the primary prevention of bleeding from esophageal varices. A randomized controlled multicentre trial [ISRCTN03215899]. BMC Gastroenterol 2003;3:22.
127. Rikkers LF, Jin G, Burnett DA, Buchi KN, Cormier RA. Shunt surgery versus endoscopic sclerotherapy for variceal hemorrhage: late results of a randomized trial. Am J Surg 1993;165:27-33.
128. Mudawi HM, Ibrahim KB. Endoscopic variceal sclerotherapy in patients with Symmers periportal fibrosis. Trop Doct 2007;37:179-81.
129. Warren WD, Zeppa R, Fomon JJ. Selective trans-splenic decompression of gastroesophageal varices by distal splenorenal shunt. Ann Surg 1967;166:437-55.
130. Hermann RE, Henderson JM, Vogt DP, Mayes JT, Geisinger MA, Agnor C. Fifty years of surgery for portal hypertension at the Cleveland Clinic Foundation. Lessons and prospects. Ann Surg 1995;221:459-66.
131. Strauss E, Sakai P, Gayotto LC, Cardoso RA, Forster S, Raia S. Size of gastroesophageal varices: its behavior after the surgical treatment of portal hypertension. Rev Hosp Clin Fac Med Sao Paulo 1999;54:193-8.
132. Lopes FG, Haddad CM. Late clinical, biochemical, endoscopic and electroencephalographic evaluation of patients with schistosomal portal hypertension treated with distal splenorenal shunt. Int Surg 1997;83:42-7.
133. Henderson JM, Millikan WJ, Warren WD. The distal splenorenal shunt: an update. World J Surg 1984;8:722-32.
135. Henderson JM. Surgery versus transjugular intrahepatic portal systemic shunt in the treatment of severe variceal bleeding. Clin Liver Dis 2006;10:599-612.
136. Warren WD, Millikan WJ Jr, Henderson JM, Abu-Elmagd KM, Galloway JR, Shires GT 3rd, Richards WO, Salam AA, Kutner MH. Splenopancreatic disconnection: improved selectivity of distal splenorenal shunt. Ann Surg 1986;204:346-55.
137. Henderson JM, Warren WD, Millikan WJ, Galloway JR, Kawasaki SE, Kutner MH. Distal splenorenal shunt with splenopancreatic disconnection. A 4-year assessment. Ann Surg 1989;210:332-41.
138. Bessa SM, Helmy I, El-Sheikh SO, Hamam SM, El Khishen MA. The distal splenorenal shunt in patients with variceal bleeding due to schistosomal hepatic fibrosis. Surg Gynecol Obstet 1987;165:143-7.
140. Da Silva LC, Strauss E, Gayotto LC, Mies S, Macedo AL, da Silva AT, Silva EF, Lacet CM, Antonelli RH, Fermanian J. A randomized trial for the study of the elective surgical treatment of portal hypertension in mansonic schistosomiasis. Ann Surg 1986;204:148-53.
141. Spina GP, Henderson JM, Rikkers LF, Teres J, Burroughs AK, Conn HO, Pagliaro L, Santambrogio R, Ascione A, Bordas JM, Brooks WS. Distal spleno-renal shunt versus endoscopic sclerotherapy in the prevention of variceal rebleeding: a meta-analysis of 4 randomized clinical trials. J Hepatol 1992;16:338-45.
142. Henderson J. Distal splenorenal shunts. In: Jarnagin W, editor. Surgery of the liver, biliary tract and pancreas. 5th ed. Philadelphia: Elsevier; 2012. pp. 1166-71.
143. Elwood DR, Pomposelli JJ, Pomfret EA, Lewis WD, Jenkins RL. Distal splenorenal shunt: preferred treatment for recurrent variceal hemorrhage in the patient with well-compensated cirrhosis. Arch Surg 2006;141:385-8.
144. Warren WD, Henderson JM, Millikan WJ, Galambos JT, Bryan FC. Management of variceal bleeding in patients with noncirrhotic portal vein thrombosis. Ann Surg 1988;207:623-34.
145. Moon SB, Jung SE, Ha JW, Park KW, Seo JK, Kim WK. The usefulness of distal splenorenal shunt in children with portal hypertension for the treatment of severe thrombocytopenia and leukopenia. World J Surg 2008;32:483-7.
146. Yoshida H, Mamada Y, Taniai N, Mineta S, Kawano Y, Mizuguchi Y, Kanda T, Tajiri T. Shunting and nonshunting procedures for the treatment of esophageal varices in patients with idiopathic portal hypertension. Hepatogastroenterology 2009;57:1139-44.
147. De Cleva R, Herman P, Pugliese V, Zilberstein B, Saad WA, Rodrigues JJ, Laudanna AA. Prevalence of pulmonary hypertension in patients with hepatosplenic Mansonic schistosomiasis-prospective study. Hepatogastroenterology 2002;50:2028-30.
148. Salam A. Distal splenorenal shunts: hemodynamics of total versus selective shunting. In: Fischer J, editor. Mastery of surgery. 6th ed. Philadelphia: Lippincott Williams & Willkins; 2012. pp. E21-E9.
149. Warren WD, Salam AA, Hutson D, Zeppa R. Selective distal splenorenal shunt: technique and results of operation. Arch Surg 1974;108:306-14.
150. Zeppa R. Splenorenal shunt. Curr Surg Tech 1977;1:5-15.
151. De Cleva R, Herman P, Pugliese V, Zilberstein B, Saad WA, Gama-Rodrigues JJ. Fathal pulmonary hypertension after distal splenorenal shunt in schistosomal portal hypertension. World J Gastroenterol 2004;10:1836-7.
152. Jenkins RL, Gedaly R, Pomposelli JJ, Pomfret EA, Gordon F, Lewis WD. Distal splenorenal shunt: role, indications, and utility in the era of liver transplantation. Arch Surg 1999;134:416-20.
153. Henderson JM, Boyer TD, Kutner MH, Galloway JR, Rikkers LF, Jeffers LJ, Abu–Elmagd K, Connor J. Distal splenorenal shunt versus transjugular intrahepatic portal systematic shunt for variceal bleeding: a randomized trial. Gastroenterology 2006;130:1643-51.
154. Henderson JM, Nagle A, Curtas S, Geisinger M, Barnes D. Surgical shunts and TIPS for variceal decompression in the 1990s. Surgery 2000;128:540-7.
155. Nagasue N, Kohno H, Ogawa Y, Yukaya H, Tamada R, Sasaki Y, Chang YC, Nakamura T. Appraisal of distal splenorenal shunt in the treatment of esophageal varices: an analysis of prophylactic, emergency, and elective shunts. World J Surg 1989;13:92-9.
156. Orozco H, Mercado MA, Granados JG, Hernandez-Ortiz J, Tielve M, Chan C, Contreras JL. Selective shunts for portal hypertension: current role of a 21-year experience. Liver Transpl Surg 1997;3:475-80.
157. Khaitiyar JS, Luthra SK, Prasad N, Ratnakar N, Daruwala DK. Transjugular intrahepatic portosystemic shunt versus distal splenorenal shunt-a comparative study. Hepatogastroenterology 1999;47:492-7.
158. Orozco H, Mercado MA. The evolution of portal hypertension surgery: lessons from 1000 operations and 50 years' experience. Arch Surg 2000;135:1389-93.
159. Rossle M, Haag K, Ochs A, Sellinger M, Noldge G, Perarnau JM, Berger E, Blum U, Gabelmann A, Hauenstein K, Langer M. The transjugular intrahepatic portosystemic stent-shunt procedure for variceal bleeding. N Engl J Med 1994;330:165-71.
160. Sanyal AJ, Freedman AM, Shiffman ML, Purdum PP, Luketic VA, Cheatham AK. Portosystemic encephalopathy after transjugular intrahepatic portosystemic shunt: results of a prospective controlled study. Hepatology 1994;20:46-55.
161. Somberg KA, Riegler JL, LaBerge JM, Doherty-Simor MM, Bachetti P, Roberts JP, Lake JR. Hepatic encephalopathy after transjugular intrahepatic portosystemic shunts: incidence and risk factors. Am J Gastroenterol 1995;90:549-55.
162. Knechtle SJ, D'Alessandro AM, Armbrust MJ, Musat A, Kalayoglu M. Surgical portosystemic shunts for treatment of portal hypertensive bleeding: outcome and effect on liver function. Surgery 1999;126:708-13.
163. Henderson JM, Warren WD. Current status of the distal splenorenal shunt. Semin Liver Dis 1983;3:251-63.
164. Millikan WJ Jr, Warren WD, Henderson JM, Smith RB 3rd, Salam AA, Galambos JT, Kutner MH, Keen JH. The Emory prospective randomized trial: selective versus nonselective shunt to control variceal bleeding. Ten year follow-up. Ann Surg 1985;201:712-22.
165. Strickland GT. Liver disease in Egypt: hepatitis C superseded schistosomiasis as a result of iatrogenic and biological factors. Hepatology 2006;43:915-22.
166. Abrantes WL, Vieira JN, Fernades AD, Coutinho AO, Rabelo GD, Dupin JB, De Oliveira MR. Embolization of the splenic artery for treatment of acute "cor pulmonale" following selective portal decompression in patients with hepatosplenic schistosomiasis and pulmonary hypertension. AMB Rev Assoc Med Bras 1981;27:110.
167. Starzl TE, Marchioro TL, Porter KA, Brettschneider L. Homotransplantation of the liver. Transplantation 1967;5:790-803.
168. Calne RY, Williams R, Dawson JL, Ansell ID, Evans DB, Flute PT, Herbertson PM, Joysey V, Keates GH, Knill-Jones RP, Mason SA. Liver transplantation in man-II, a report of two orthotopic liver transplants in adult recipients. Br Med J 1968;4:541-6.
169. Adam R, Karam V, Delvart V, O'Grady J, Mirza D, Klempnauer J, Castaing D, Neuhaus P, Jamieson N, Salizzoni M, Pollard S. Evolution of indications and results of liver transplantation in Europe. A report from the European Liver Transplant Registry (ELTR). J Hepatol 2012;57:675-88.
170. Starzl TE, Demetris AJ, Van Thiel D. Medical progress: liver transplantation. N Engl J Med 1989;321:1014-99.
171. Hashikura Y, Makuuchi M, Kawasaki S, Matsunami H, Ikegami T, Nakazawa Y, Kiyosawa K, Ichida T. Successful living-related partial liver transplantation to an adult patient. Lancet 1994;343:1233-4.
172. Arulraj R, Neuberger J. Liver transplantation: indications and general considerations. In: Jarnagin W, editor. Surgery of the liver, biliary tract and pancreas. 5th ed. Philadelphia: Elsevier; 2012. p. 1663.
173. Lo CM, Fan ST, Liu CL, Lai CL, Chan JK, Ng IO, Fung A, Wong J, Wei W. Adult-to-adult living donor liver transplantation using extended right lobe grafts. Ann Surg 1997;226:261-9.
174. Orloff MJ, Orloff MS, Girard B, Orloff SL. When is liver transplant indicated in cirrhosis with bleeding varices? Transplant Proc 2001;33:1366.
176. Rikkers LF. Definitive therapy for variceal bleeding: a personal view. Am J Surg 1990;160:80-5.
177. Hoare M, Gelson WT, Davies SE, Curran M, Alexander GJ. Hepatic and intestinal schistosomiasis after orthotopic liver transplant. Liver Transpl 2005;11:1603-7.
178. Henderson JM, Gilmore GT, Hooks MA, Galloway JR, Dodson TF, Hood MM, Kutner MH, Boyer TD. Selective shunt in the management of variceal bleeding in the era of liver transplantation. Ann Surg 1992;216:248-55.
179. Iwatsuki S, Starzl TE. Liver transplantation in the management of bleeding esophageal varices. Baillieres Clin Gastroenterol 1992;6:517-25.
180. Vincenzi R, Neto JS, Fonseca EA, Pugliese V, Leite KR, Benavides MR, Cândido HL, Porta G, Miura IK, Pugliese R, Danesi VB, Guimarães TC, Porta A, Kondo M, Carone E, Chapchap P. Schistosoma mansoni infection in the liver graft: the impact on donor and recipient outcomes after transplantation. Liver Transpl 2011;17:1299-303.
181. Pannegeon V, Masini JP, Paye F, Chazouillères O, Girard PM. Schistosoma mansoni infection and liver graft. Transplantation 2005;80:287.
182. Jacobs DL, Rikkers LF. Indications and results of shunt operations in the treatment of patients with recurrent variceal hemorrhage. Hepatogastroenterology 1990;37:571-4.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Doush WMA, Elzein JA. Surgical outcomes of distal splenorenal shunt or liver transplantation in treatment of schistosomal refractory variceal bleeding. J Unexplored Med Data 2018;3:2. http://dx.doi.org/10.20517/2572-8180.2017.18
AMA Style
Doush WMA, Elzein JA. Surgical outcomes of distal splenorenal shunt or liver transplantation in treatment of schistosomal refractory variceal bleeding. Journal of Unexplored Medical Data. 2018; 3: 2. http://dx.doi.org/10.20517/2572-8180.2017.18
Chicago/Turabian Style
Doush, Wael Mohialddin Ahmed, Juhaina Ahmed Elzein. 2018. "Surgical outcomes of distal splenorenal shunt or liver transplantation in treatment of schistosomal refractory variceal bleeding" Journal of Unexplored Medical Data. 3: 2. http://dx.doi.org/10.20517/2572-8180.2017.18
ACS Style
Doush, WMA.; Elzein JA. Surgical outcomes of distal splenorenal shunt or liver transplantation in treatment of schistosomal refractory variceal bleeding. J. Unexplored. Med. Data. 2018, 3, 2. http://dx.doi.org/10.20517/2572-8180.2017.18
About This Article
Copyright
Data & Comments
Data

 Cite This Article 7 clicks
Cite This Article 7 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.